TUBERCULOSIS
Tuberculosis
remains a major infectious disease both worldwide and in the United States. The
pathogen responsible for most cases, Mycobacterium tuberculosis, infects
one third of the world’s population, and it is responsible for over 2 million
deaths per year. The incidence of this disease is declining in the United
States at a rate of approximately 3% to 6% per year; however, tuberculosis
remains an important cause of mortality and morbidity among those in immunocom promised states, especially those coinfected with the
human immunodeficiency virus (HIV).
Although a majority of those infected with Mycobacterium
tuberculosis develop disease restricted to the lungs, a recent survey of
cases in the United States found that 19% had only extrapulmonary disease, whereas
6% had combined pulmonary and extrapulmonary disease. Of those with
extrapulmonary disease, 6.5% had urogenital involvement. In addition, the
relative proportion of extrapulmonary cases appears to be increasing: despite a
steady decline in the number of new pulmonary tuberculosis cases, there has
been little change in the number of new extrapulmonary cases. Worldwide,
urogenital involvement is even more common, occurring in up to 40% of
extrapulmonary cases. There is evidence, however, that even this number may be
an underestimate; in one autopsy study, 73% of patients with pulmonary
tuberculosis were found also to have a renal focus.
Urogenital tuberculosis affects men twice as often as
women, and the average age at presentation is approximately 40 years old. HIV
infection is also a major risk factor not only for active tuberculosis in
general, but also for extrapulmonary spread and reactivation.
PATHOPHYSIOLOGY
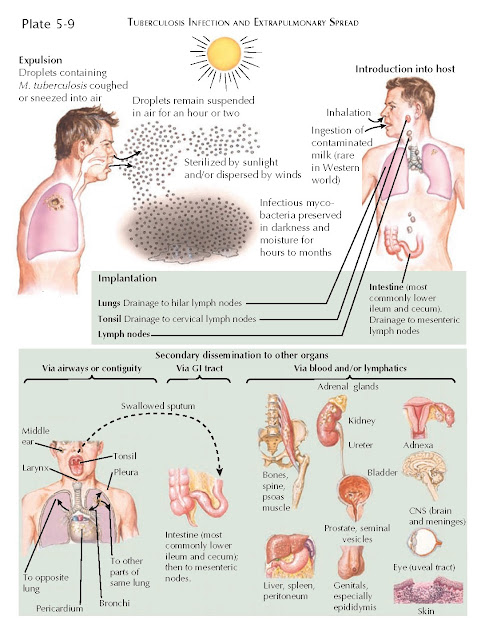
Upon inhalation of airborne bacilli, patients may experience
a primary, usually silent, infection that involves formation of granulomas in
the pulmonary alveoli. During this initial phase, lymphatic and then
hematogenous seeding of distant organs such as the kidneys and reproductive organs can occur. In rare instances, wide and uncontrolled
dissemination of mycobacteria may lead to miliary tuberculosis, which can also
involve the kidneys (see separate section later).
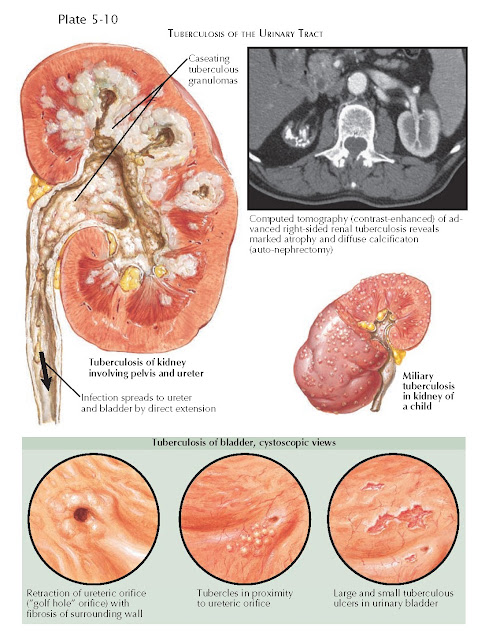
In the urinary system, the renal cortex is the usual
site of primary infection. After initial seeding, the disease course is
indolent. Indeed, bacilli can remain latent within granulomas for decades or
more, both in the kidneys and elsewhere. Reactivation can occur because of a
decline in immunity because of age, disease, or malnutrition. Reactivation of
renal tuberculosis may lead to further granuloma formation, parenchymal
cavitation, papillary necrosis, calcification, and, in rare cases, tuberculous
interstitial nephritis.
As the renal disease becomes advanced, it may spread
to the rest of the urinary system by direct extension. In the ureter,
strictures and calcification may occur. In the bladder, ulceration and fibrosis
may occur, leading to wall contraction and a decrease in storage capacity. Fibrosis
adjacent to the ureteric orifice may cause it to become retracted and assume a
“golf hole” appearance.
Genital disease may occur either because of
hematogenous spread or contiguous extension from the urinary system.
PRESENTATION AND DIAGNOSIS
The symptoms of genitourinary tuberculosis can be very
nonspecific. Patients often complain of urinary frequency and may, in some
cases, experience gross hematuria or flank pain. Some patients may also have
constitutional symptoms, including fever and weight loss.
About 90% of patients will have abnormal urinalysis,
which may reveal positive leukocyte esterase, hematuria, proteinuria, and low
urine pH. About 1 in 10 patients will have only frank hematuria, whereas up to
half have microscopic hematuria. As Mycobacterium tuberculosis does not
convert urinary nitrates, dipstick is often negative for nitrites.
The classic urinary finding found in up to one quarter of patients is sterile pyuria, where urine contains numerous white
blood cells but no bacterial growth is seen on standard cultures. Of note,
bacterial growth does not necessarily rule out renal tuberculosis, since
secondary bacterial infection is common.
If sterile pyuria is seen, the differential diagnosis
also includes chlamydial urethritis, pelvic inflammatory disease,
nephrolithiasis, or renal papillary necrosis. If constitutional symptoms and
hematuria are present, a malignancy of the urinary or genital system should
also be suspected.
A physician may suspect urogenital tuberculosis if the
patient has risk factors for tuberculosis exposure, a history of a positive
purified protein derivative (PPD) test, a history of immunocompromise, and
constitutional symptoms. It is not uncommon, however, for physicians to
prescribe antibacterial treatment at first presentation. A lack of positive
urine cultures, no response to antimicrobials, or recurrent episodes of
cystitis in the setting of suggestive risk factors should raise a suspicion of
mycobacterial infection and prompt further evaluation. A radiograph may reveal areas
of focal calcification in the kidneys and the lower genitourinary tract.
Ultrasonography may reveal calcification, hypoechoic renal abscesses, and
shrunken kidneys. CT may demonstrate calcifications, renal scarring and
cavitation, papillary necrosis, strictures of the collecting system, and
diminution of renal function. Imaging of the thorax should also be performed to
rule out concomitant pulmonary or spinal infection. Many patients will have
evidence of prior pulmonary infection, and up to 30% to 40% will be found to
have active pulmonary disease.
Once tuberculosis is suspected, the bacillus can be
identified in urine by acid-fast staining. Because sensitivity is low, multiple
early morning midstream voided specimens are often collected to increase
detection power. Culture on either liquid broth or solid Löwenstein-Jensen
medium remains the gold standard for diagnosis, and it also has the advantage
of providing information about mycobacterial drug susceptibilities.
Although culture sensitivity can be as high as 80%,
results can take as long as 6 to 8 weeks. For that reason, polymerase chain
reaction (PCR) studies may be performed to detect M. tuberculosis in
urine earlier in the clinical course. The advantage of this test is not only
its high sensitivity (up to 95%), but also its fast turnaround time, with
results often obtained within 24 hours.
In situations when there is a high clinical suspicion
for tuberculous disease, but microbiologic testing is not definitive, more
invasive methods of diagnosis, such as percutaneous tissue biopsy, may be
required. Histopathologic examination of the obtained tissue reveals caseating
granulomas.
In immunocompetent patients, an intradermal tuberculin
test (PPD) can be performed as an additional screen for tuberculosis exposure;
however, it is not helpful in diagnosis of the disease because results have
been reported as positive in only 60% of patients with urinary tuberculosis.
Interpretation of tuberculin test results should follow standard cut-off
values: 5 mm or more for immunocompromised patients, 10 mm or more for patients
at high risk (inmates, health-care workers, long-term care facility residents,
intravenous drug users, and immigrants), and 15 mm or more for patients without
any risk factors. A positive tuberculin test result indicates prior exposure
but is not diagnostic of active disease. In patients with advanced HIV or other
immunocompromising conditions, tuberculin test results may be negative
secondary to anergy and should not be used to rule out tuberculosis.
TREATMENT
The treatment of urogenital tuberculosis involves a
combination of antituberculous therapy and surgical resection, where possible,
of local disease. Consultation with experts should be sought, given not only
the complexity of the disease course but also the potential adverse effects
associated with treatment.
Pending the return of culture and sensitivities, the
patient should be initiated on a four-drug regimen that includes isoniazid,
ethambutol, rifampin, and pyrazinamide. If cultures indicate the organism is
sensitive to isoniazid, rifampin, and pyrazinamide, the ethambutol can be
stopped. After 8 weeks, pyrazinamide is usually stopped, because it has an
early bactericidal effect.
The duration of therapy with the remaining two agents depends on the patient’s immune status. Therapy may also need to be adjusted if infection occurs in an
area with high levels of multidrug-resistant or extensively drug-resistant
tuberculosis.
PROGNOSIS
In the developed world, tuberculosis is rarely a cause
of chronic kidney disease. Preservation of renal function, however, depends on
early detection to limit renal parenchymal destruction. In developing
countries, where diagnosis and treatment are more likely to be delayed,
permanent loss of renal function is more common.
MILIARY TUBERCULOSIS
In addition to being the site of locally reactivating
granulomas, as described previously, the kidneys may rarely be involved in the
disseminated disease known as miliary tuberculosis.
Miliary tuberculosis results from widespread
hematogenous dissemination of tuberculous bacilli after invasion of the
pulmonary circulation. It may occur during the time of primary infection or at
reactivation, and it is often associated with the extremes of age and other
conditions that compromise the immune system. Patients have more pronounced
constitutional systems and extensive pulmonary disease. Overwhelming systemic
illness may overshadow the effects of renal involvement. The workup of miliary
tuberculosis includes acid-fast bacillus smears, culture, PCR, and histopathologic
examination of affected tissues (e.g., bone marrow, lymph nodes, liver). If the
kidney is affected, numerous granulomatous lesions may be present throughout
the cortex and, less commonly, the medulla. On microscopic examination these
granulomas reveal central caseous necrosis.
Rapid diagnosis of military tuberculosis is essential,
and treatment with the combination regimen described above should be promptly initiated.