ECHOCARDIOGRAPHY
Echocardiography is a highly reproducible, safe, and widely available noninvasive imaging technique integral to the practice of modern clinical cardiology. With the use of high-frequency ultrasound to image cardiac and great vessel structure and blood flow, this method provides definitive anatomic and hemodynamic information crucial to the diagnosis and management of patients with a wide range of cardiac and vascular conditions. Although often considered a mature imaging technique, the technology and its applications continue to improve.
FIG 9.1
Transducer positions in
echocardiographic examination.
|
FIG 9.2
Principles of Doppler
echocardiography. |
IMAGING METHODS
AND CLINICAL APPLICATIONS
Transthoracic
Echocardiography
A comprehensive transthoracic echocardiographic examination (TTE)
includes the acquisition of standard two-dimensional (2D) and M-mode views of
the intrathoracic structures complemented by continuous-wave and pulsed-wave
spectral Doppler data and color flow Doppler imaging. Current commercial
echocardiographic imaging systems also have tissue harmonic imaging capability
that helps to enhance endocardial definition in patients with technically difficult
TTE windows. In addition, tissue Doppler imaging (TDI), which is analogous to
pulsed-wave Doppler assessment of blood flow velocity, is used to measure
longitudinal myocardial motion. When combined with a comprehensive TTE
examination, TDI can yield clinically useful information regarding diastolic
ventricular function and cardiac filling pressures. Small, lightweight, and
highly portable ultrasound systems are also available for bedside TTE imaging.
Commonly referred to as “handheld” TTE devices, these instruments possess
limited capability compared with standard echocardiographic equipment, but this
technology continues to evolve. These devices are widely used for the rapid
triage of patients in emergency department and intensive care unit settings.
Transthoracic 2D echocardiography is the foundation of the clinical
echocardiographic examination. Tomographic images are obtained usually from
four standard imaging “windows” on the chest wall, defined by the transducer
position and image plane (Fig. 9.1). These are the parasternal, apical,
subcostal, and suprasternal positions. It provides a reliable, portable, and
reproducible evaluation of cardiac chamber sizes, myocardial thickness,
ventricular contractile performance, valvular structure and function, the
pericardium, and the great vessels. Doppler echocardiographic assessment of
the direction and velocity of blood flow within the heart and great vessels is
valuable for the detection and quantification of obstructive lesions and
valvular regurgitation (Fig. 9.2).
M-mode echocardiography was the first application of ultrasound in
cardiology. It provides both high spatial and temporal (time-related)
resolution. Hence, it is especially valuable in the evaluation of mitral and
aortic valve motion in dynamic and fixed left ventricular outflow obstruction,
in the timing of mitral valve closure in aortic regurgitation, and in the
assessment of pericardial disease.
Although coronary arteries cannot be reliably imaged by TTE, the method is nevertheless valuable in the
assessment of known or suspected coronary artery disease (CAD).
Echocardiographic evidence of segmental ventricular contractile dysfunction can
be used to screen for acute or chronic ischemic myocardial injury or
infarction, secondary to CAD. However, the diagnosis of CAD is not established
by segmental wall motion abnormalities because these can also be caused by
cardiac trauma, myocarditis, and infiltrative myocardial diseases. In addition,
multivessel CAD can cause globally decreased ventricular contraction without
segmental wall motion abnormalities, a circumstance that generally requires
further evaluation.
TTE is used for the initial diagnostic evaluation and follow-up of
patients with congenital and valvular heart disease, including the assessment
of right ventricular systolic pressure and pulmonary arterial hypertension.
Anatomic information about the nature of a congenital defect and its
hemodynamic consequences, including the direction and magnitude of intracardiac
shunts and estimation of pulmonary and systemic blood flow, can be estimated by
2D and Doppler techniques (Fig. 9.3).
 |
FIG 9.3 Coarctation of the aorta.
Transthoracic 2D echocardiography provides a comprehensive picture of the valvular, subvalvular, and annular structures, and when 2D echocardiography is combined with Doppler ultrasound techniques, obstructive gradients can be accurately measured and the cross-sectional valve area can be estimated. Regurgitant valvular lesions can be accurately quantified by color flow Doppler imaging. Clinical decisions regarding medical therapy and operative intervention for patients with valvular disease are usually based on TTE 2D and Doppler echocardiographic data, supplemented by information from cardiac catheterization.
TTE is the primary tool for evaluating the presence and hemodynamic
consequences of pericardial effusion. Two-dimensional imaging and a
comprehensive Doppler examination can reliably identify patients with
pericardial effusion and tamponade pathophysiology. TTE-guided
pericardiocentesis can reduce procedural complications and improve therapeutic
results. A thickened pericardium and typical hemodynamic alterations can alert
the clinician to the diagnosis of pericardial con- striction, but magnetic
resonance imaging and catheterization are usually needed for full evaluation.
Analysis of Doppler-measured ventricular inflow velocities and TDI can be
useful in differentiating between pericardial constriction and infiltrative
cardiomyopathy.
Transesophageal Echocardiography
A transesophageal echocardiographic examination (TEE) requires an ultrasound probe placed into the esophagus, posterior to the heart. Because of the decreased distance between the transducer and the heart, as well as the absence of interference from bone and lung tissue, the signal-to-noise ratio is more favorable with TEE than with TTE, and higher frequency transducers can be used to improve resolution. There- fore TEE image quality is generally superior to that of TTE, particularly for posterior structures, including the pulmonary veins, left atrium, interatrial septum, and mitral valve. TEE is most commonly applied in the evaluation for suspected patent foramen ovale (PFO), atrial septal defects (ASDs), quantification of valvular regurgitation, valvular vegetations, left atrial or atrial appendage thrombus, and aortic disease. TEE is being used with increasing frequency in clinical cardiac electrophysiology before elective cardioversion and invasive procedures, including ablative therapy for atrial fibrillation (Fig. 9.4).
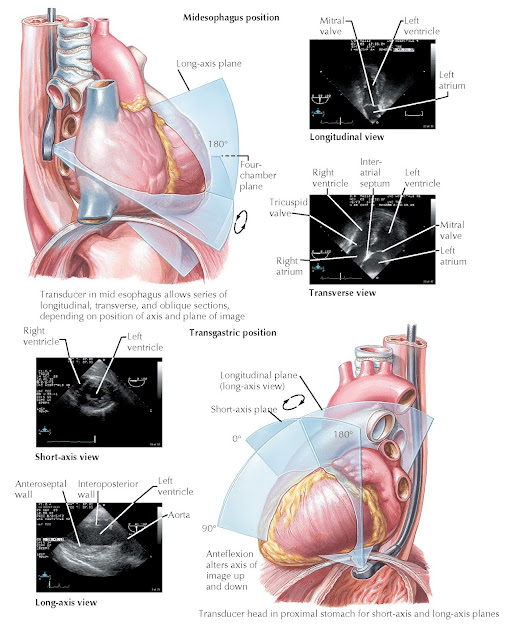 |
FIG
9.4 Transesophageal
echocardiography.
Stress Echocardiography
Exercise and pharmacological stress echocardiography enables evaluation of the heart at rest
and during stress. The clinical usefulness of stress echocardiography depends
on acquisition of high-quality TTE images of the left ventricle, in multiple
planes, at maximal cardiac workload. With exercise stress, patients must be
highly motivated not only to reach, but ideally exceed, a target heart rate,
because cardiac workload falls rapidly with cessation of exercise.
Repositioning of the subject and immediate poststress image acquisition usually
requires 30 to 60 seconds, and test sensitivity falls when echocardiographic
data are recorded at less than maximal workload. For these reasons, equivocal
test results are fairly common with exercise stress (unless a recumbent bike is
used). This is rarely a problem with pharmacological stress test (usually
dobutamine) because patient repositioning is not necessary, and maximal cardiac
workload can be maintained while image acquisition is completed.
Pharmacological stress also has a technical advantage in that patients are not
moving during the study; sequential images can be recorded as cardiac workload
is gradually increased, and respiratory interference at peak stress is not a
limiting factor.
Stress echocardiography is an accurate, noninvasive approach to detect the presence and extent of CAD. A
stress-induced segmental wall motion abnormality usually indicates
flow-limiting CAD. In addition to providing a useful approach for detecting
obstructive CAD, stress echocardiography can be used to assess an area of
myocardium at risk, for detection of myocardial viability, in risk
stratification after myocardial infarction, and for evaluation of the results
of coronary revascularization. Stress echocardiography is especially useful in
detecting CAD in patients after heart transplantation, in those being
considered for renal transplantation, and for preoperative evaluation of
individuals under- going vascular surgery. Exercise echocardiography is invaluable
to the assessment of exercise-induced pulmonary hypertension, especially in the
setting of mitral valve disease and evaluation of dynamic left ventricular
outflow tract gradients in patients with hypertrophic obstructive
cardiomyopathy. Low-dose dobutamine stress echocardiography plays an important
role in the evaluation of asymptomatic or low gradient aortic stenosis (Fig.
9.5).
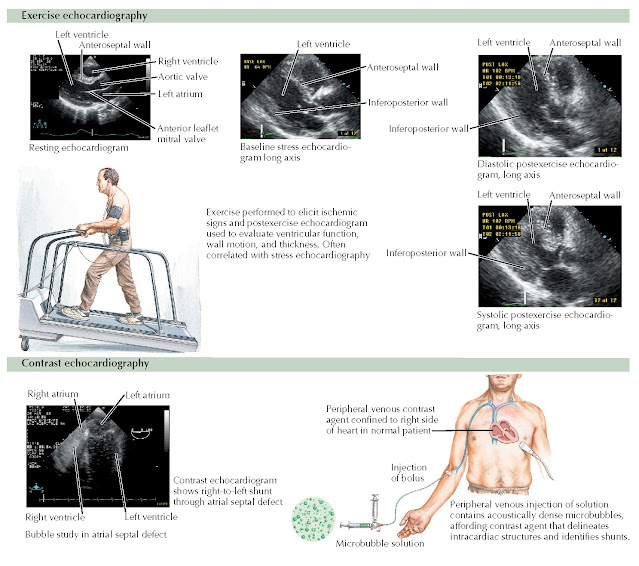 |
FIG
9.5 Exercise and contrast
echocardiography.
Contrast Echocardiography
Contrast echocardiography is now widely used to detect intracardiac and
intrapulmonary shunts, to augment Doppler velocity signals, and to enhance
endocardial border definition. Intravenous injection of agitated normal saline
is most often used for opacification of the right heart, shunt detection, and
augmentation of tricuspid regurgitant jets to allow more accurate estimation of
right ventricular systolic pressure. Commercially available contrast agents,
termed “microbubbles,” are made of a high-molecular-weight gas encapsulated in
a shell of phospholipid or protein. Modifications of the microbubble shell and
gas properties have resulted in improved stability of these agents as they pass
through the pulmonary circulation after intravenous injection, and high-quality
imaging of the left heart chambers can be reliably obtained. Myocardial
perfusion imaging with contrast echocardiography is not routinely used for
clinical purposes.
Strain Imaging
Strain is an assessment of myocardial deformation with strain rate being
the rate of myocardial deformation in time. It can be obtained from TDI or from
the speckle tracking method. TDI can be used even in poor echocardiographic
windows. Speckle tracking allows for assessment of longitudinal (global
longitudinal strain), radial, and circum-ferential strain. There is an
expanding role for strain imaging for the assessment of segmental wall motion
abnormalities, cardiac dyssynchrony analysis, right ventricular function,
infiltrative cardiomyopathies (e.g., amyloidosis) and monitoring of oncology
patients on cardiotoxic drugs such as adriamycin or trastuzumab (Herceptin) (Fig.
9.6).
 |
FIG 9.6 Abnormal
myocardial strain pattern in a patient with amyloidosis.
|
FIG 9.7 Three-dimensional
echocardiogram demonstrating a cleft in the posterior leaflet of the mitral valve. |
Intravascular Ultrasound and Intracardiac Echocardiography
The development of intravascular ultrasound (IVUS) and intracardiac
echocardiography (ICE) techniques has extended the application of
echocardiography and bridged traditional boundaries between noninvasive and
invasive imaging methods. IVUS uses a miniaturized transducer on the end of a
flexible, steerable catheter that is inserted into arteries, which allows in
vivo ultrasound imaging of vascular anatomy from the inside of the artery. ICE
relies on a catheter-like ultrasound probe that can be advanced to the right
heart chambers via the femoral vein and inferior vena cava, and it also crosses
the interatrial septum. ICE probe technology has evolved rapidly and is capable
of high-resolution 2D echocardiography and a full complement of Doppler
imaging modalities.
Intracoronary IVUS is commonly used in cardiac catheterization laboratories to delineate atherosclerotic
plaque morphology, lesion length, and
obstruction severity when standard coronary angiographic and pressure data are ambiguous. Intracoronary IVUS can help guide
percutaneous coronary intervention and stent implantation, and aid in the diagnosis of in-stent
restenosis. ICE is also often used to monitor noncoronary interventional
procedures in interventional electrophysiology and cardiac catheterization
laboratories. ICE has proven useful for direct visualization of the pulmonary
veins and left atrial appendage during invasive ablation procedures for atrial
fibrillation. In addition, ICE is now used to assist with guidance of
radiofrequency catheter ablation of atrial arrhythmias in the right side of the
heart. ICE augments fluoroscopy through improving visualization of landmarks,
ensuring endocardial contact, and assisting with transseptal puncture.
This technique is also useful in the prompt detection of procedural complications, including intracardiac
thrombus formation, pericardial effusion, and pulmonary vein obstruction.
Three-Dimensional Echocardiography
Three-dimensional (3D) echocardiography, via either a transthoracic or
transesophageal approach, can provide improved definition of spatial
relationships between normal and abnormal cardiac structures and eliminate the
need for cognitive reconstruction of image planes currently required for
interpretation of standard 2D images.
Three-dimensional echocardiography has the potential to provide more accurate and reliable measurements of
cardiac chamber dimensions and
function. This is especially true and probably most important when dealing with
complex shapes such as the right ventricle or aneurysmal left ventricle because
quantification by 2D methods, which rely on geometric assumptions about shape,
are less accurate. Significant advances in ultrasound, electronic, and computer
technology have made real-time–rendered 3D images more practical and
potentially valuable in clinical practice. There is evidence to support the use
of 3D echocardiography for quantification of left ventricular mass, volume,
and ejection fraction, as well as in the measurement of the mitral valve area
in patients with mitral stenosis (Fig. 9.7).
Interventional
Echocardiography
Over the past decade, percutaneous catheter–based interventions of
various structural heart disorders have expanded dramatically. This has led to
an emerging new field of interventional echocardiography, especially with the
addition of 3D imaging. Interventions involving percutaneous balloon mitral
valvuloplasty, and percutaneous repair of the mitral valve (MitraClip procedure) and the tricuspid valve heavily
rely on both 2D and 3D TEE support.
TEE is also useful in the cardiac catheterization laboratory to assist with
transseptal puncture and for optimal percutaneous placement of closure devices
in patients with PFO or ASD and left atrial appendage closure. Transcatheter
aortic valve replacements have usually relied on TEE guidance; however, many
centers are currently performing at least some cases with conscious sedation
and TTE imaging. Although these procedures have expanded into use in the lower
risk populations, TEE imaging will continue to play an important role in ensuring optimal outcomes.
LIMITATIONS
Although modern echocardiography imaging systems are sophisticated
multimodality devices, echocardiography remains an operator-dependent
technique. High-quality echocardiographic imaging requires a solid foundation
of training in cardiac anatomy, cardiovascular physiology, and
pathophysiology. A working knowledge of ultrasound physics, as well as considerable technical skill,
expertise, and patience of the physician or technician obtaining images, is
essential. Even in trained hands, image acquisition is limited by obesity,
chronic obstructive pulmonary disease, and patient discomfort; chest wall
injuries or recent surgery can make TTE particularly challenging. Suboptimal
images may be seen in up to 10% to 15% of all patients
undergoing echocardiography. Because of this difficulty, contrast media have
been used more widely to enhance endocardial definition.
TEE is limited in many clinical circumstances. Patients must be
physically able, well-oriented, and sufficiently cooperative to follow simple
commands to successfully swallow the ultrasound probe. Although generally well tolerated, TEE carries
risks related to sedation and esophageal intubation; complications include
esophageal perforation and aspiration
of gastric contents.



.webp)
_Page_049.jpg)
