LEGG-CALVÉ-PERTHES DISEASE
Legg-Calvé-Perthes disease is defined as idiopathic avascular necrosis of the epiphysis of the femoral head (capital femoral epiphysis) and its associated complications in a growing child. It is a common, but poorly understood hip disorder.
The disease develops more often in boys than girls (4 or 5 : 1). It can
occur between 2 and 12 years of age (mean age, 7 years); and when the
involvement is bilateral, the changes usually appear in one hip at least 1 year
earlier than in the other. If the child is older than 12 years of age at the
time of clinical onset, the disorder is not considered true Legg-Calvé-Perthes
disease but rather adolescent avascular necrosis, which has a poor prognosis
similar to that of the adult form.

LEGG-CALVÉ-PERTHES DISEASE: PATHOGENESIS
PREDISPOSING FACTORS
Genetic Aspects
The incidence of Legg-Calvé-Perthes disease is 1% to 20% higher in
families of involved children, although there is no consistent pattern of
inheritance. Studies in England have indicated that affected children are more
likely than normal children to have low birth weight, abnormal birth
presentation (breech and transverse presentations), and older parents. The
disease is also more prevalent in later-born children (particularly the third
to the sixth child).
The disorder occurs more frequently in Asian, Eskimo, and Central
European populations, whereas the incidence is decreased in blacks, Australian
aborigines, American Indians, and Polynesians.
The English studies have also demonstrated a higher than normal
incidence of minor congenital genitourinary anomalies (e.g., renal
abnormalities, inguinal hernias, and undescended testes) in affected children
as well as in their first-degree relatives.
Abnormal Growth and Development
Legg-Calvé-Perthes disease may be a manifestation of an unknown systemic
disorder rather than an isolated abnormality of the hip joint. The bone age of
affected children is typically 1 to 3 years lower than their chronologic age.
As a consequence, affected children are usually shorter than their peers, and
the shortness of stature, although slight, persists into adulthood.
Disproportionate growth, abnormalities in skeletal growth and
maturation, and elevated serum levels of somatomedin have been demonstrated.
Affected children are typically smaller in all dimensions except head
circumference, and their limbs have disproportionately small distal segments.
The relationship between growth abnormalities, serum somatomedin, and ischemia
of the epiphysis of the femoral head remains obscure. However, these findings
support the concept of an underlying systemic disorder.
Environmental Factors
Although the effect of environment on the incidence is not clear, a
large number of affected children in England are from lower socioeconomic
groups. Whether this reflects dietary or en ironmental influences or a
combination is not clear.
ETIOLOGY AND PATHOGENESIS
The etiology of Legg-Calvé-Perthes disease is not yet understood, but it
is accepted that the avascular necrosis is due to an interruption of the blood
supply to the epiphysis of the femoral head, especially the contributions from
the superior and inferior retinacular arteries. Current etiologic theories
include trauma to the retinacular vessels, vascular occlusion secondary to
increased intracapsular pressure from acute transient synovitis, venous
obstruction with secondary intra-epiphyseal thrombosis, vascular irregularities
(congenital or developmental), and increased blood viscosity resulting in
stasis and decreased blood flow.
Although the cause remains unclear, numerous studies have delineated the
pathogenesis of Legg-Calvé-Perthes disease. Initially, an ischemic episode of
unknown etiology occurs, rendering most, if not all, of the epiphysis avascular
(see Plate 2-31).
Endochondral ossification in the preosseous epiphyseal cartilage and
growth plate ceases
temporarily, while the articular cartilage, which is nourished by synovial
fluid, continues to grow. This results in the radiographic appearance of a
widened medial cartilage (joint) space and a smaller ossification center in the
involved hip. This is the first radiographic manifestation, and it precedes any
change in the density of the epiphysis. At this stage, the marrow space of the
epiphysis is necrotic.
Revascularization of the structurally intact but avascular epiphysis
occurs from the periphery as new capillaries recanalize the previous vascular
channels. Resumption of endochondral ossification within the epiphysis begins
peripherally and progresses centrally. With the ingrowth of capillaries,
osteoclasts and osteoblasts cover the surface of the avascular subchondral
cortical bone and the central trabecular bone. New bone is deposited on the
avascular bone, producing a net increase in bone mass per unit area; this
accounts for the increased density of the epiphysis that is apparent on
radiographs taken in early stages of the disease. The deposition of new
trabecular bone and resorption of avascular bone occur simultaneously. In the
subchondral area, bone resorption exceeds new bone formation. A critical point
is reached during resorption when the subchondral area becomes biomechanically
weak and therefore susceptible to a pathologic fracture. Up to this point, the
disease process is clinically silent and asymptomatic. The continuation of this
“potential” form of Legg-Calvé-Perthes disease or the development of the “true”
form depends on whether a sub-chondral fracture occurs.
In the potential form of the disease, a subchondral fracture does
not occur because the stresses and shearing forces acting on the revascularized
epiphysis of the femoral head do not exceed the strength of the weakened
subchondral area. The reossification process continues uninterrupted, with
ultimate resumption of normal growth and development. Thus, there is no
epiphyseal resorption, no extrusion or subluxation of the femoral head, and no
potential for deformity. The child remains asymptomatic and retains a good
range of motion in the hip joint. The subchondral area eventually regains its
normal strength and stability, and a “head-within-a-head” is visible on
radiographs. The head-within-a-head represents a growth arrest line that
outlines the ossification center at the time of the initial infarction.
In the true form of the disease, the strength of the weakened
subchondral area is exceeded and a pathologic subchondral fracture occurs (see
Plate 2-31). The magnitude of stress or trauma necessary to produce such a
fracture is difficult to quantitate and appears to vary both with the degree of
preexisting weakness and the applied shearing forces. In most cases, the
fracture seems to result from normal vigorous activity rather than from a
specific injury. The painful subchondral fracture heralds the clinical onset of
true Legg-Calvé- Perthes disease, and only the true form produces the typical
clinical and radiographic features and requires 2 to 4 years,
or even longer, for complete healing to occur.
 |
| LEGG-CALVÉ-PERTHES DISEASE: PHYSICAL EXAMINATION |
Changes in Epiphysis
The subchondral fracture characteristically begins in the anterolateral
aspect of the epiphysis near the growth plate, because this area receives the
greatest concentration of stress during weight bearing. The pathologic fracture
extends superiorly and posteriorly until it reaches areas where the strength of
the remaining sub- chondral bone exceeds the shearing forces acting on the
femoral head. There is minimal, if any, extension of the subchondral
fracture after the initial fracture. The reasons for this are not clear, but
presumably the resulting pain causes the child to be less active, thereby
reducing the stress on the femoral head.
The revascularized trabecular bone beneath the sub-chondral fracture
undergoes a second episode of local ischemia secondary to trabecular collapse
and occlusion of the ingrowing capillaries. This second ischemic episode,
mechanical in origin, involves either part or all of the epiphysis,
depending on the extent of the sub-chondral fracture. The structural stability
of the epiphysis is lost; the ingrowth of new capillaries is impeded by the
obliteration of the vascular channels and the presence of fractured bone (both
cortical and trabecular) and marrow debris. Consequently, the entire area is
slowly revascularized, with resorption of the fibro- osseous tissue, by a
process termed creeping substitution. In this reparative process, the avascular
bone is slowly resorbed from the periphery of the area of the second infarction
and replaced by vascular fibrous tissue that, in turn, is eventually replaced
by primary trabecular bone.
During the process of creeping substitution, the femoral head, while not
soft in the physical sense, can be molded into a round or flat shape by the
forces acting on it. This remodeling property, or biologic plasticity, lasts
until subchondral reossification begins. Potential deformities may be caused by
the different rates of growth within the femoral head—areas not undergoing
resorption grow faster than the involved area. The combined factors of pressure
and asymmetric growth result in a potential for extrusion and subluxation of
the femoral head and eventual deformity. Thus, true Legg-Calvé-Perthes disease
is actually a complication of avascular necrosis.
Secondary alterations in the growth plate and metaphysis also occur and
can lead to further disturbances in endochondral ossification and growth in the
proximal femur.
Changes in Growth Plate
Because the blood supply to the growth plate comes from the epiphyseal
side, the two ischemic episodes also produce ischemic changes in the growth
plate. The chondrocyte columns become distorted with some loss of their
cellular components; they do not undergo normal ossification, which results in
an excess of calcified cartilage in the primary trabecular bone.
 |
| LEGG-CALVÉ-PERTHES DISEASE: PHYSICAL EXAMINATION (CONTINUED) |
Changes in Metaphysis
Four types of metaphyseal changes have been noted: presence of adipose
tissue, osteolytic lesions (well-circumscribed areas of fibrocartilage),
disorganized ossification, and extrusion of the growth plate. Whereas only
adipose tissue changes are detected early in the disease, osteolytic lesions
are seen in the later stages. When these fibrocartilaginous lesions are in
contact with the growth plate, the normal architecture of the growth plate is
lost and the lesions appear on radiographs as cysts. In the areas without
osteolytic lesions, ossification is disorganized and bars, or columns, of unossified
cartilage appear to “stream” or “flow” down into the metaphysis. Necrosis of
bone is not seen in the metaphysis. In some severely deformed femoral heads,
the growth plate extrudes down the sides of the femoral neck.
The changes in the growth plate and metaphysis ultimately alter the
growth in length of the proximal femur and produce the short, thick femoral
neck (coxa vara) and enlarged femoral head (coxa magna) typically seen in
Legg-Calvé-Perthes disease. The greater trochanter, being uninvolved, continues
to grow and may eventually rise above the level of the femoral head. The
combination of a short femoral neck and a high greater trochanter is considered
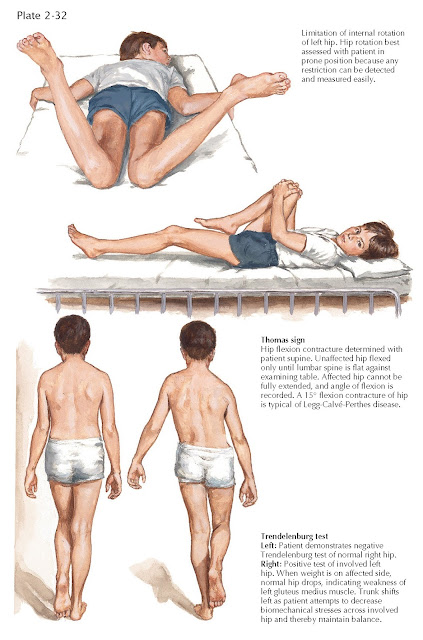
“functional” coxa vara. The performance of the hip abductor (gluteus medius)
muscles is disturbed, with a resultant limp or Trendelenburg gait and a
positive Trendelenburg test (see Plate 2-32). The short femoral neck also
produces a lower limb-length discrepancy of 1 to 2 cm.
CLINICAL MANIFESTATIONS
The pertinent early findings include antalgic gait, muscle spasm and
restricted hip motion, atrophy of the proximal thigh, and short stature. A
small percentage of children have a history of trauma that is usually mild.
Nevertheless, such trauma may be sufficient to produce the pathologic subchondral
fracture.
Initial symptoms are mild and intermittent pain in the anterior thigh or
a limp, or both. Although many children do
not complain of pain, on close questioning most admit to mild pain either in
the anterior thigh or the knee. The onset of pain may be acute or insidious.
Referred pain from the hip to the anterior thigh or knee must be considered.
Because the child’s initial symptoms are typically mild, parents frequently do
not seek medical attention for several weeks after clinical onset, or longer.
Antalgic gait is noted when the patient shortens the time of weight
bearing on the involved limb during walking to reduce discomfort. Pain from the
irritable hip can also cause reflex inhibition of the hip abductor muscles with
a resultant positive Trendelenburg test, a common early sign (see Plate 2-32).
Muscle spasm is best detected by the “roll” test, a painless test
that reveals any guarding or muscle spasm (secondary to irritability of the hip
joint), especially when the involved limb is rolled inward (see Plate 2-32).
Once the child’s confidence is gained, the hip can usually be
examined more thoroughly to determine the complete range of motion. Mild
limitation of motion, particularly abduction and internal rotation, is the
typical finding. This may be best elucidated by noting asymmetry in both
abduction and internal rotation. There may also be limitation of extension, as
evidenced by a mild hip flexion contracture (Thomas sign), as well as deep
tenderness over the anterior aspect of the hip. Disuse atrophy of the
proximal thigh muscles is a consequence of prolonged hip irritability and the
resultant limitation of motion. The atrophic thigh is usually 2 to 3 cm
smaller, especially during the early symptomatic phases. As the symptoms
subside, the atrophy resolves. Short stature due to delayed bone age is
another typical finding in affected children. The patient’s bone age can be
determined with the Greulich and Pyle atlas. Results of laboratory tests are
normal, except for an occasionally abnormal erythrocyte sedimentation rate, which may be
slightly elevated (30 to 40 mm/hr).

STAGES OF LEGG-CALVÉ-PERTHES DISEASE
RADIOGRAPHIC EVALUATION
Routine radiographic assessment is essential for diagnosis and for
determining progression of the disease, sphericity of
the femoral head, possibility of epiphyseal extrusion or collapse, and response
to treatment. Arthrography is a useful adjunct, especially in the setting of
the operating room to best define the true sphericity, or lack thereof, of the
femoral head. Magnetic resonance imaging (MRI) can also be helpful in rare
cases, and radionuclide bone scanning currently plays a very limited role.
The entire disease process can usually be assessed from plain
anteroposterior and Lauenstein frog-leg radiographs of the pelvis (both hips).
Extrusion and subluxation of the femoral head can be measured on these
radiographs using the Wiberg center-edge angle. An extrusion index developed by
Green and associates has been demonstrated to be prognostically significant.
Sphericity of the femoral head in the reossification and healed stages is
currently best determined by the Mose circle criteria. In this technique, a
transparent template with concentric circles at 2-mm intervals, placed on both
anteroposterior and frog-leg radiographs, is centered over the femoral head to
measure both the sphericity and diameter of the femoral head. If the sphericity
is equal in both projections, the hip is rated “good.” A variance of up to 2 mm
is rated “fair,” whereas a variance of 3
mm or more is rated “poor.” The good and fair ratings are considered
satisfactory results, whereas poor ratings are unsatisfactory. Sphericity may
improve with growth and development if the healed femoral head remains well
contained in the acetabulum.
Computerized methods are being investigated to allow better objective
quantification of hip joint architecture and for plotting changes in
configuration that occur with time.
Early in the resorption stage, arthrography may be required to assess
the sphericity of the articular surface of the femoral head. The contour of the
partially resorbed ossification center of the epiphysis may not reflect the
contour of the articular surface, and range of motion in the hip is usually the
best indicator of potential femoral head deformity. Only “questionable” hips
require arthrography.
Bone scans have largely been replaced by MRI. MRI is helpful in defining
epiphyseal infarction and the contours of the femoral head, both of which are
prognostically significant. Like radionuclide bone scans, MRI does not
correlate with the extent of epiphyseal involvement.
LEGG-CALVÉ-PERTHES DISEASE: LATERAL PILLAR CLASSIFICATION
STAGES OF DISEASE
Radiographic evaluation has determined five distinct stages of
Legg-Calvé-Perthes disease, which represent a continuum of the disease process.
Growth Arrest
This stage occurs immediately after the initial ischemic episode in the
femoral head, when endochondral ossification of the preosseous cartilage
ceases. During this avascular phase, which may last 6 to 12 months, there is a
slight but progressive difference in the size (height and width) of the
involved epiphysis and that of the opposite normal hip. The joint space also
appears to be wider because of the continued growth of the articular cartilage.
These relatively small differences (1 to 3 mm) are visible and measurable on an
anteroposterior radio- graph of the pelvis. Toward the end of this stage,
epiphyseal density increases. During this stage, which is only potential
Legg-Calvé-Perthes disease, the disease is clinically silent and asymptomatic.
Subchondral Fracture
The subchondral fracture initiates true Legg-Calvé- Perthes disease.
Radiographic visibility of the fracture varies with the age of the patient at
clinical onset and the extent of epiphyseal involvement. The duration varies
from an average of 3 months in children 4 years of age or younger to 812 months
in children 10 years or older.
Resorption
In this stage, also called fragmentation or necrosis, the necrotic bone
beneath the subchondral fracture is gradually and irregularly resorbed. This
process produces the radiographic appearance of fragmentation because the bone
is resorbed and replaced by vascular fibrous tissue
(creeping substitution) and later by primary bone. The resorption phase lasts 6
to 12 months and is longest when there is extensive epiphyseal involvement or
when the child is 10 years of age or older at clinical onset. This phase is
usually complete 12 to 17 months after clinical onset.
Reossification
During the healing, or reossification, stage, ossification of the primary
bone begins irregularly in the subchondral area and
progresses centrally. Eventually, the newly formed areas of bone coalesce and
the epiphysis progressively regains its normal strength. Reossification takes 6
to 24 months.
Healed Stage
The healed, or residual, stage signals the complete ossification of the
epiphysis of the femoral head, with or without
residual deformity.
There have been many classification systems developed to describe the
disease process. Most such as the Catterall and Salter-Thompson classification
systems are helpful retrospectively but have had little prognostic value. The
lateral pillar classification system is currently the most widely used and
valuable classification system because it has been shown to have some prognostic
significance.
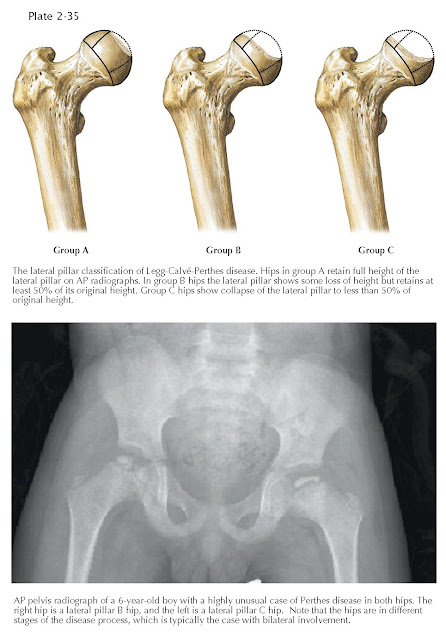
The lateral pillar classification system developed by Hering and
colleagues separates diseased hips into three groups (A, B, and C) on the basis
of the remaining height of the lateral third of the femoral head. Group A hips
have maintenance of 100% of the lateral pillar height. These have the best
prognosis long term and are least likely to extrude. Group B hips have
decreased lateral pillar height but have at least 50% of the lateral pillar
height remaining. Group C hips have less than 50% of the lateral pillar height
remaining and have an almost uniformly poor prognosis.

LEGG-CALVÉ-PERTHES DISEASE: CONSERVATIVE MANAGEMENT
PROGNOSIS
The short-term prognosis for patients with Legg-Calvé-Perthes disease
focuses on femoral head deformity at the completion of the healing stage. The long-term
prognosis involves the potential for secondary osteoarthritis of the hip in
adulthood.
Deformity of Femoral Head
The ultimate goal of treatment is a spherical femoral head at the
completion of growth. Six factors determine the potential for femoral head
deformity.
1.
Sex of patient. In general,
the outcome is less favorable in girls than in boys. Involvement of the femoral
head is often more extensive in girls; and because they mature earlier than
boys, there is less remaining skeletal growth from the time of clinical onset
and consequently less opportunity for epiphyseal remodeling.
2. Age at clinical onset. The older the
child at clinical onset, the less favorable the prognosis, particularly in
children 10 years of age and older. This may also be related to the reduced
remaining skeletal growth and potential for femoral head remodeling in older
children.
3. Extent of epiphyseal involvement. More extensive
involvement is correlated with a poorer prognosis.
4. Containment of femoral head. Extrusion,
subluxation, or asymmetric growth of the femoral head increases the stress
concentrated on it during weight bearing. The ability to maintain the femoral
head well within the acetabulum with appropriate treatment is a significant
factor for a favorable prognosis.
5. Persistent loss of motion. This is
usually due to either muscle spasm (adductors or iliopsoas muscle), muscle
contractures, anterolateral extrusion or subluxation of the femoral head, or a
com- bination thereof. The loss of motion prevents adequate remodeling of the
femoral head by the acetabulum.
6. growth plate. When
involvement of the epiphysis is extensive (lateral pillar group C), the growth
plate may be sufficiently damaged to cause premature closure. This can result
in asymmetric growth and inadequate remodeling that contributes to femoral head
deformity, greater trochanteric overgrowth (functional coxa vara), and a lower
limb-length discrepancy.
Late Degenerative Osteoarthritis
The incidence of late degenerative osteoarthritis depends on residual
deformity of the femoral head and the patient’s age at clinical onset. The risk
is directly correlated with the extent of residual deformity. Three types of
congruency between the femoral head and the acetabulum have been classified:
spherical congruency, aspherical congruency, and aspherical incongruency.
Spherical congruency is not associated with osteoarthritis,
whereas aspherical congruency predisposes to mild-to-moderate osteoarthritis in
late adult-hood. Patients with aspherical incongruency usually develop
degenerative osteoarthritis before age 50.
Studies also show that the incidence of osteoarthritis of the hip in
adults with deformed femoral heads is negligible in patients 5 years of age or
younger at the time of clinical onset, 38% in patients 6 to 9 years of age, and
100% in patients 10 years of age or older. Aspherical incongruency, a
predisposing factor for osteoarthritis, is also more likely to develop in
children who are older at the time of clinical onset.
Thus, of the two significant factors in the longterm prognosis, only
femoral head deformity may be preventable, or at least altered, by appropriate
treatment.
FEMORAL VARUS DEROTATIONAL OSTEOTOMY
TREATMENT
The only justification for treatment is prevention of femoral head
deformity and secondary osteoarthritis. When indicated, treatment should
interfere as little as possible with the child’s psychological and physical
development.
The four basic goals of treatment are to eliminate hip irritability,
restore and maintain a good range of hip motion, prevent femoral head extrusion
and sub-luxation, and attain a spherical femoral head on healing.
Elimination of Hip Irritability
After the subchondral fracture, the synovium becomes inflamed and the
hip irritable. The associated pain and muscle spasm lead to the restriction of
motion followed by muscle contractures, especially of the adductor and
iliopsoas muscles, and possible anterolateral extrusion or subluxation of the
femoral head. Elimination of this irritability is always the first objective
and is usually accomplished by rest and scheduled anti-inflammatory
medications. Non–weight bearing for brief periods may also help the symptoms of
irritability, and crutches or other aids can be helpful if the child is able to
use them.
Restoration and Maintenance of Motion Generally,
satisfactory range of motion in the hip returns as the hip irritability is
eliminated, although residual stiffness may persist in some children.
Physical therapy with passive and active range-of-motion exercises helps to
restore motion, but gentle progressive-abduction traction, especially at night,
is occasionally required. To maintain hip motion, a program consisting of
abduction and internal rotation stretching exercises may be helpful.
Regardless of the sphericity of the femoral head, almost all children
with lateral pillar group C involvement show a slight but persistent loss of
abduction and internal rotation due to mild coxa magna.
Prevention of Femoral Head Collapse Extrusion or
subluxation of the femoral head increases the risk of epiphyseal collapse and
subsequent deformity. Radiographic evidence of extrusion is therefore a prognostic
factor and an indication for treatment.
Attainment of Spherical Femoral Head
This goal requires a full understanding of the pathogenesis and
prognostic factors associated with deformity of the femoral head as well as the
appropriate management techniques.
CONCEPTS OF CONTAINMENT
Until the 1960s, treatment for Legg-Calvé-Perthes disease was complete
and prolonged bed rest—with or without traction or
abduction of the involved limb—and the use of so-called weight-relieving
devices. All children were treated, and treatment often lasted 2 to 4 years.
Containment techniques have been devised to permit weight bearing while
redirecting the compressive forces on the femoral head to assist in the healing
and remodeling process. The currently accepted forms of
management range from observation to surgery.
Appropriate treatment of all children who are younger than 6 years of
age at clinical onset regardless of the extent of epiphyseal involvement is by
observation only, provided there is no limitation of hip motion and no
subluxation. Observation is also appropriate for children 6 years of age or
older with lateral pillar A and some lateral pillar B involvement who have a
good range of hip motion and no radiographic evidence of femoral head extrusion
or collapse.
Intermittent Symptomatic Treatment
Temporary or periodic bed rest and abduction stretching exercises can be
used in conjunction with observation. Hip irritability with a temporary
decrease in motion often recurs during the subchondral fracture and resorption
phases. If these symptoms persist and there is no radiographic evidence of
femoral head extrusion, rest and protected weight bearing for 1 to 2 weeks
sometimes may be necessary. Two or three recurrent episodes of irritability may
indicate the need for a short period (1 to 2 months) of nonsurgical containment
to decrease the risk of extrusion. Radiographs should be taken at 2- to 4-month
intervals to ensure that the irritability is not due to early deformity of the
femoral head.
 |
| INNOMINATE OSTEOTOMY |
Definitive Early Treatment
Nonsurgical or surgical containment of the femoral head early in the
disease is indicated in children 6 years of age or older at clinical
onset—possibly in girls 5 years of age or older—who have lateral pillar group B
or B/C border involvement or when femoral head extrusion is seen on the
weight-bearing anteroposterior radiograph.
Use of containment techniques requires a good-to-full range of hip
motion (especially abduction), no residual irritability, and a round or almost
round femoral head. Containment methods, whether nonsurgical or surgical,
appear to increase satisfactory results (most good and fair) by 16% to 20%
compared with no treatment or natural history.
Nonsurgical containment refers to the use of
abduction casts (Petrie) or occasionally an orthosis to abduct the involved
limb and redirect the femoral head within the acetabulum (see Plate 2-36). The
Petrie cast fixes the lower limbs in 30 to 40 degrees of abduction with an
approximate 5-degree internal rotation. The cast provides continuous
containment because it cannot be removed by either the child or the parents. Disadvantages
include stiffness of the knee and ankle joints with adaptive articular changes,
significant restriction in ambulation, frequent need for change and repair, and
excessive weight. Petrie casts are now reserved for management after surgical
adductor lengthening to improve and maintain motion in abduction, therefore
“containing” the joint.
Abduction braces are lighter and less cumbersome than casts, but they
are quite expensive. Also, because they are removable, compliance may not be
consistent. Their use is now largely historical. The Atlanta Scottish Rite
Children’s Hospital brace and Salter stirrup provided temporary nonsurgical
containment.
Surgical containment has three major
advantages: (1) the period of restriction is less than 2 months,
after which the child may gradually return to full activity; (2) the
femoral head containment is permanent; and (3) the permanent improvement in
containment continues to enhance remodeling of the healed femoral head long
after the active phase of disease is over. Surgery does not alter the length of
the disease process or provide a cure, but it does provide satisfactory results
in the great majority of patients.
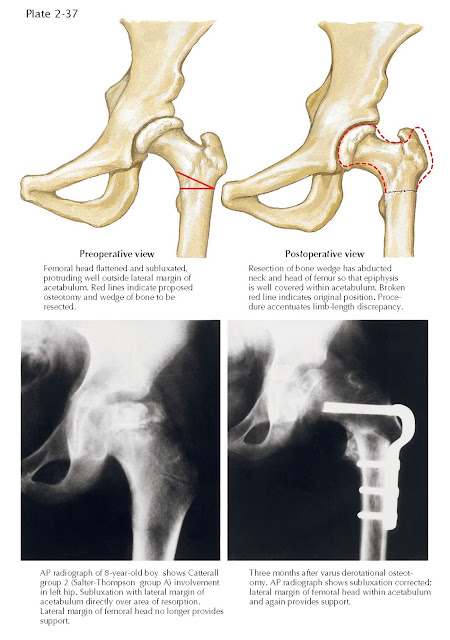
Treatment with femoral varus derotational osteotomy usually involves a
varus angulation of the proximal femur, with or without rotation, to redirect
the femoral head into the acetabulum (see Plate 2-37). The varus
angulation should be no greater than 110 degrees but should allow containment
of the epiphysis of the femoral head within a vertical (Perkin’s) line drawn on
the radiograph at the lateral margin of the acetabulum; some surgeons also
recommend 10 to 15 degrees of internal rotation of the proximal segment. The
osteotomy is usually held securely with threaded screws and a side plate or
blade plate. Femoral osteotomy, while a technically less demanding procedure
than innominate osteotomy,
produces some inherent problems, mainly the increase in lower limb-length
discrepancy, potential coxa vara, and Trendelenburg gait. In addition, the
metal fixation device should be removed and there is a small risk of fracture
of the proximal femur through the screw holes. The limb shortening associated
with femoral osteotomy usually resolves in younger children and in patients who
achieve satisfactory results.
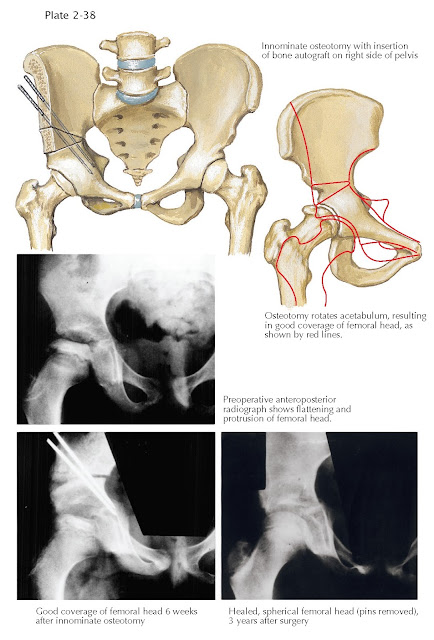
In 1962, Salter began to treat older children with more severe forms of
the disease with innominate osteotomy (see Plate 2-38), which is a technically
more difficult procedure than femoral osteotomy. However, its advantages
include better anterior and lateral coverage of the femoral head, no further
shortening of the femoral neck (coxa breva), no increase in limb-length
discrepancy (it actually lengthens the lower limb by about 1 cm), and
improvement of the Trendelenburg gait. Also, removal of fixation devices is
easier and there is no risk of fracture of the proximal femur. The triple
innominate osteotomy has also been utilized for containment. The Salter
innominate osteotomy can be combined with a proximal femoral osteotomy for
additional containment. Although even more technically demanding than the
Salter innominate osteotomy, the triple innominate osteotomy provides
significantly more containment when needed, without the disadvantages of the
proximal femoral procedure (varus, limp, and Trendelenburg gait and implant
removal).
 |
| INNOMINATE OSTEOTOMY (CONTINUED) |
Late Surgical Management for Deformity
If a significant deformity prevents reduction of the femoral head into
the acetabulum or remodeling after treatment with standard containment methods,
an alternative must be considered. Several surgical procedures at least
partially correct the various existing deformities, thereby alleviating the
associated symptoms. These are salvage procedures that can alleviate symptoms
but do not favorably alter the natural history of the disease.
Proximal femoral valgus osteotomy is employed when the radiograph
demonstrates that hip joint congruency is better when the extended hip is
adducted. The biomechanics of the hip joint are improved by this procedure in
which the greater trochanter is moved distally as well as laterally, thus
enhancing the strength of the abductor muscles as well as increasing the range
of abduction of the lower limb.
Premature closure of the epiphysis of the femoral head can occur in
advanced forms of the disease, resulting in shortening of the femoral neck and
progressive overgrowth of the greater trochanter. Advancing the greater
trochanter distally and laterally relieves muscle pain and decreases or
eliminates the characteristic Trendelenburg gait. Lateral displacement of the
greater trochanter can also decrease the pressure between the femoral head and
the acetabulum and may minimize the risk of late degenerative osteoarthritis.
In addition to the proximal femoral valgus osteotomy, a Chiari
osteotomy or shelf osteotomy may produce improved coverage of the
femoral head and reduction of symptoms. These procedures are, again, salvage
procedures that are designed to reduce the short-term symptoms, but
unfortunately do not seem to alter the natural history of severe
Legg-Calvé-Perthes disease, which is ultimately
early arthrosis of the joint.
Late Surgical Management for
Degenerative Osteoarthritis
Significant degenerative osteoarthritis in adults is usually managed by
total hip replacement.
The prognosis for children with Legg-Calvé-Perthes disease is much
better now than in the past. Active treatment is not always required, and many
patients need only careful observation or intermittent symptomatic treatment. A
variety of nonsurgical and surgical containment
techniques are available that produce equally good long-term results. When
surgical treatment is required, it restricts the child for relatively short
period of time, thus reducing the potential for psycho- logical problems.
Further studies will concentrate more on the etiology of
Legg-Calvé-Perthes disease than on better treatment. Only greater understanding
of the disease can provide the means for eliminating it or significantly altering its
course.



.webp)
