Neurotransmitters, Receptors And Their Pathways
Neurotransmitters and synaptic
function
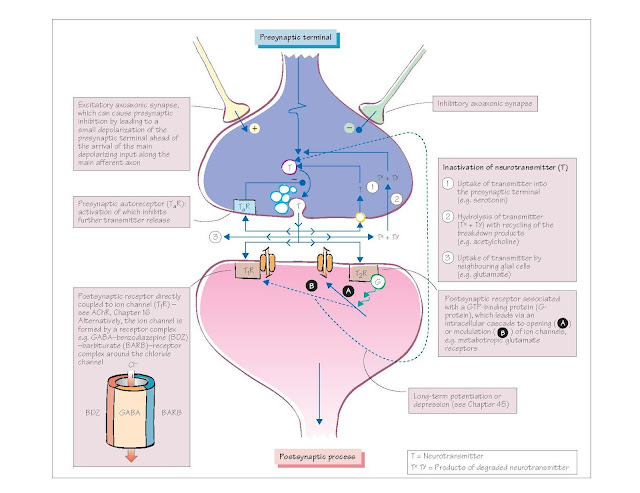
The neurotransmitter released at a
synapse interacts with a specific protein in the postsynaptic membrane, known
as a receptor. At some synapses the neurotransmitter also binds to a presynaptic
autoreceptor that regulates the amount of transmitter that is released.
Receptors are usually specific for
a given neurotransmitter, although several different types of that receptor may
exist. In some cases co-released neurotransmitters can either modulate the
binding of another neurotransmitter to its receptor or act synergistically on a
common single ion channel (e.g. the γ-aminobutyric acid [GABA]–benzodiazepine–barbiturate
receptor).
Receptors for specific
neurotransmitters are either coupled directly to ion channels (T1R
on figure, e.g. acetylcholine receptors (AChR); see Chapter 16) or to a
membrane enzyme (T2R). In these latter instances the
binding of the neurotransmitter to the receptor either opens an ion channel via
an intracellular enzyme cascade (e.g. cyclic adenosine monophosphate [cAMP] and
G-proteins) or indirectly modulates the probability of other ion channels
opening in response to voltage changes (neuromodulation). These
receptors therefore mediate slower synaptic events, unlike those receptors
directly coupled to ion channels that relay fast synaptic information.
The activated receptor can only
return to its resting state once the neurotransmitter has been removed either
by a process of enzymatic hydrolysis or uptake into the
presynaptic nerve terminal or neighbouring glial cells. Even then there are
often intermediate steps in the process of returning the receptor and its
associated ion channel to the resting state. At some synapses the affinity and,
ultimately, the number of receptors is dependent on the previous activity of
the synapse. For example, at catecholaminergic synapses the receptors become
less sensitive to the released transmitter when the synapse is very active – a
process of desensitization and down-regulation. This process involves a
decrease in the affinity of the receptor for the transmitter in the short term,
which goes on in the long term to an actual decrease in the number of
receptors. The converse is true with synapses that are rarely activated (super-sensitivity
and up-regulation), and in this way synaptic activity is modulated by its
ongoing activity.
In addition, at some synapses the
activation of the postsynaptic receptor–ion channel complex can modulate the
long-term activity of the synapse, either by affecting the presynaptic release
of neurotransmitter or the postsynaptic receptor response – a process known as
either long-term potentiation (LTP) or long-term depression (LTD)
depending on the actual change in synaptic efficacy over time (see Chapters 45
and 49). Therefore the state, number and types of receptor for a specific
neurotransmitter as well as the presence of receptors to other
neurotransmitters are all important in determining the extent of synaptic
activity at any given synapse.
Diversity and anatomy of neurotransmitter
pathways
The nervous system employs a large
number of neurotransmitters, which can be divided into groups (see also Chapter
19).
Excitatory amino acids
These represent the main excitatory
neurotransmitters in the central nervous system (CNS) and are important at most
synapses in maintaining ongoing synaptic activity. The main excitatory amino
acid is glutamate, which acts at a number of receptors (which are
defined by the agonists that activate them). The inotropic receptors
consist of the N-methyl-D-aspartate (NMDA) and non-NMDA
receptors, and the former receptor with its associated calcium channel may
be important in the generation of LTP (see Chapter 45), excitotoxic cell death
(see Chapter 60) and possibly epilepsy (see Chapter 61).
A separate group of G-protein
associated glutamate receptors, the metabotropic receptors, respond on
activation by initiating a number of intracellular biochemical events that
modulate synaptic transmission and neuronal activity. These receptors may
underlie long-term depression in the hippocampus.
Inhibitory amino acids
The major CNS inhibitory
neurotransmitters are GABA, which is present throughout the CNS, and glycine
which is predominantly found in the spinal cord. Abnormalities of GABA
neurones may underlie some forms of movement disorders as well as anxiety states
and epilepsy (see Chapters 59 and 61). While mutations in the glycine receptor
have now been linked to some forms of hyperexplexia – a condition
in which there is an excessive startle response, such that any stimulus induces
a stiffening of the body with collapse to the ground without any impairment of
consciousness.
Monoamines
The monoaminergic systems of the
CNS originate from small groups of neurones in the brainstem, which then
project widely to all areas of the CNS. They are found at many other sites
within the body, including the autonomic nervous system (ANS; see Chapter 3).
In all locations they bind to a host of different receptors and thus can have
complex actions including a role in depression, schizophrenia, cognition and
movement control (see Chapters 41, 42, 47, 57 and 58).
Acetylcholine
This neurotransmitter is widely
distributed throughout the nervous system, including the neuromuscular junction
(see Chapter 16) and ANS (see Chapter 3). Therefore, many agents have been
developed that target the different cholinergic synapses in the periphery and
which are used routinely in surgical anaesthesia. Several disease processes can
affect the peripherally located cholinergic synapses (see Chapter 16), while
secondary abnormalities in the central cholinergic pathways may be important in
dementia of the Alzheimer type and Parkinson’s disease (see
Chapters 42 and 60).
Neuropeptides
These neurotransmitters, of which
there are many different types, are found in all areas of the nervous system
and are often co- released with other neurotransmitters. They can act as conventional
neurotransmitters as well as having a role in neuromodulation (e.g. pain
pathways; see Chapters 32 and 38).


.webp)

