Reproductive Genetics
Chromosomes
Human chromosomes are complex
structures consisting of deoxyribonucleic acid (DNA), ribonucleic acid (RNA)
and protein. Each single helix of DNA is bound at each end with a telomere and
has a centromere somewhere along the length of the chromosome. The telomere
protects the ends of the chromosome during DNA replication. Telomere shortening
is associated with aging. The centromere is the site at which the mitotic
spindle will attach and is necessary for proper segregation of chromosomes
during cell division. The centromere divides the chromosome into two arms,
identified as p (petit) for the short arm and q for the long arm. The
centromere can be positioned anywhere along the arm of the chromosome and its
location has been used to group like chromosomes together as central
(metacentric), distal (acrocentric) or others (submetacentric). The length of
the chromosome plus the position of its centromere are used to identify individual
chromosomes within the 22 pairs of autosomes and one pair of sex chromosomes.
Chromosomes are numbered in descending order of size; 1 is the largest. The
only exception to this rule is chromosomes 21 and 22; 22 is larger than 21.
Because of the historical convention of associating Down syndrome with trisomy
21, this chromosome pair was not renamed when the size difference became
apparent.
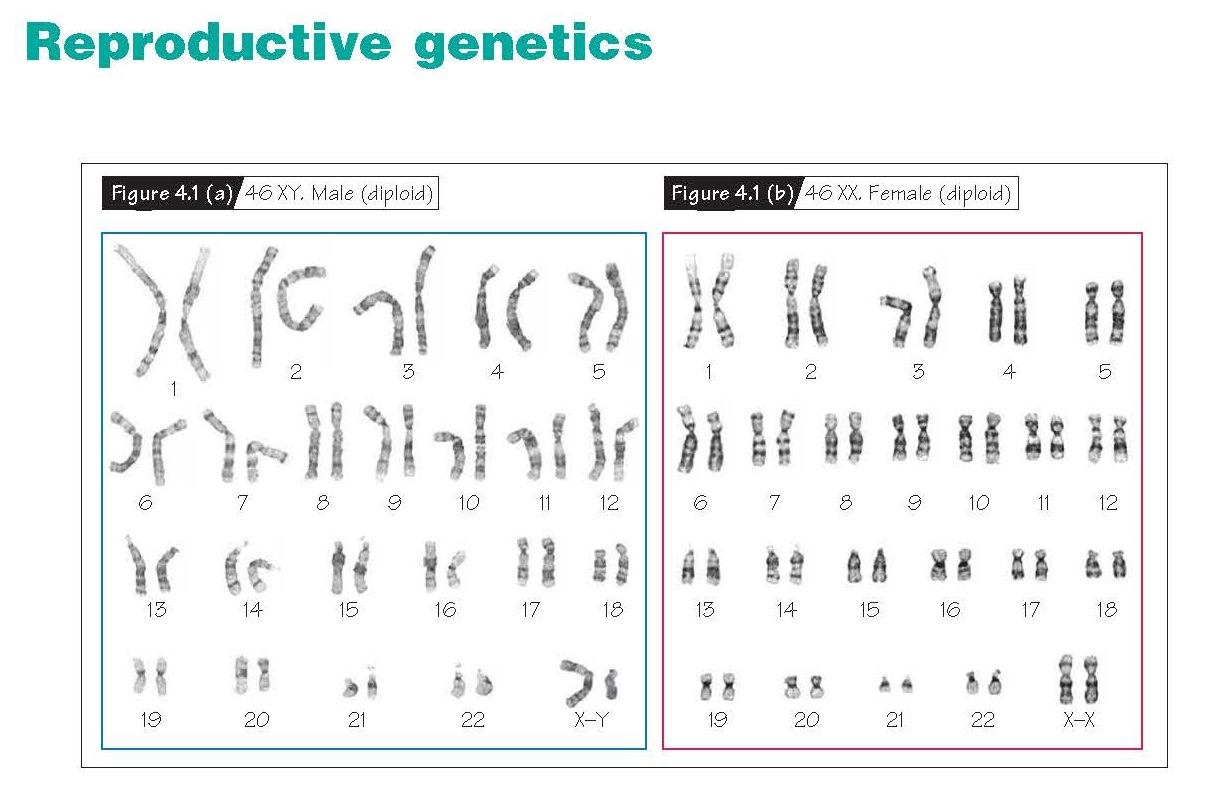
A karyotype is a display
of chromosomes ordered from 1 to 22 plus the sex chromosomes, with each
chromosome oriented so that the p arm is on top. Females have a 46XX karyotype
and males a 46XY karyotype (Fig. 4.1a and b).
Mitosis and meiosis
These are two distinct types of
cell divisions, with several common features. The first is the need to
duplicate the entire chromosome content of the cell prior to division. Both
also use the cell machinery of the parent cell to make the DNA, RNA and new
proteins that will participate in the cell division. Finally, both processes
rely on using the mitotic spindle to separate the chromosomes into the two
poles of the cell that are destined to become the progeny of that cell. Mitosis and meiosis differ in that duplicated
chromosomes behave differently after DNA replication (Fig. 4.2). In mitosis,
there is no difference in total chromosome content between parent and daughter
cells; in meiosis, the chromosome number of the daughter cells is eventually
reduced from 46 to 23, which is necessary to convert the diploid germ cell
precursors originating in the embryo into haploid (1n) germ cells. These
haploid germ cells will produce a new diploid organism at fertilization.
Meiosis promotes exchange of genetic material through chromatid crossing over;
mitosis does not.
During the interphase preceding
cell division, the DNA for each chromosome is duplicated to 4n. Thus,
each chromosome consists of two identical chromatids joined at the
centromere.
In mitosis, the chromosomes first
shorten and thicken and the nucleoli and nuclear membrane break down (prophase).
During metaphase, a mitotic spindle forms between the two centrioles of
the cell and all chromosomes line up on its equator. The centromere of each
chromosome splits and one chromatid from each chromosome migrates to the polar
ends of the mitotic spindle (anaphase). In telophase, new
nucleoli and nuclear membranes form, the parent cell divides into two daughter
cells and the mitotic spindle is disassembled. Two genetically identical cells
now exist in place of the parent cell. Mitosis is a non-sexual or vegetative
form of reproduction.
Meiosis involves two sequential
cell divisions, again beginning with the 4n DNA produced in interphase.
In prophase of the first division (prophase 1), several specific and
recognizable events occur. In the leptotene stage, the chromosomes
become barely visible as long thin structures. Homologous pairs of chromosomes
then come to lie side by side along parts of their length, forming tetrads (zygotene
stage). The chromosomes thicken and shorten, much as they do in mitotic
prophase (pachytene stage); however, the pairing that occurred in the
zygotene stage allows synapsis, crossing-over and chromatid
exchange to happen. In the diplotene/diakinesis stage, the chromosomes
shorten even more. The paired homologous chromosomes show evidence of the
crossing-over and chromatid exchange, displaying characteristic
chiasmata that join the chromosome arms. Loops and unusual shapes within the
chromosomes may be apparent at this stage. In metaphase 1 of meiosis,
the nuclear membrane breaks down and the joined pairs of homologous chromosomes
line up at the equator of the spindle apparatus. One of each pair of homologous
chromosomes then moves to each end of the cell along the spindle (anaphase 1).
Nuclear membranes may then form, yielding two haploid daughter cells with 23 2n
chromosomes in telophase 1. In the second meiotic division, these
haploid cells divide as if in mitosis. This second division produces four
haploid cells each containing 23 1n chromosomes. Unlike the cells
produced in mitosis, these daughter germ cells are genetically unique and
different from the parent cells because of the genetic exchanges that took
place in the diplotene stage. Haploid germ cells participate in sexual
reproduction in which a sperm cell and oocyte come together to form a new
diploid zygote.
While the sequence of events in
meiosis during spermatogenesis and oogenesis is basically the same, there are
several important differences. In the prepubertal male, primordial germ cells
are arrested in interphase. At puberty, these cells are reactivated to enter
rounds of mitoses in the basal compartment of the seminiferous tubule. These
reactivated cells are known as spermatogonial stem cells. From this reservoir
of stem cells, early spermatogonia emerge and divide several times again to
produce a “clone” of sperma togonia with identical genotypes. All the sperma togonia
from the clone then entermeiosis
1 and 2 toproduce unique haploid sperm.
New stem cells are constantly entering the spermatogenic cycle (Chapter 8) and thus the sperm supply is constantly
renewing itself. Because of the relatively short time for spermatocytes to
progress through meiosis and because of the tremendous competition among
spermatozoa to reach the single oocyte within the female tract, fertilization
of an egg by an aneuploid sperm is far rarer than the converse.
In contrast to the testis, the
ovary of a female at birth contains all the germ cells it will ever have. These
oocytes remain arrested in prophase 1 of meiosis until the LH surge at
ovulation initiates met- aphase 1. Thus, the duplicated genetic
material within the oocyte exists paired with its homologous chromosome for
10–50 years before the cell is called upon to divide. For this reason alone,
oocytes are much more prone to chromosome abnormalities than are sperm.
Non-disjunction
This is the failure of a
chromosome pair to separate during meiosis, and can occur at either meiosis 1
or 2. When a single chromosome is involved, the aneuploid zygote is either monosomic
or trisomic for the chromosome pair that failed to divide properly. With
the exception of monosomy X or Turner syndrome, monosomic embryos are uni-formly
miscarried (Chapter 36). Most trisomic fetuses are also miscarried; only three
(trisomy 13, 18 and 21) are reported among live births. Those that survive to
birth are likely mosaics that carry non affected
cell lineages. If all the chromosomes are present in multiples other than 2n,
the embryo or fetus is polyploid.
Imprinting
Although it is critical that the
zygote has 2n chromosomes, it is also important that one set of chromosomes
comes from each parent. Dermoid cysts and hydatidiform moles (gestational
trophoblastic disease; Chapter 45) each have all 46 chromosomes from a single
parent. Cytogenetic studies of these entities have shown the importance of
imprinting in early embryonic development. Imprinting is the process by which
specific genes are methylated so that they can no longer be transcribed. Normal
embryonic development requires that one set of genes be maternally imprinted
and a second paternally. Otherwise, important steps in development will not
occur and the zygote cannot form normally. For instance, two sets of maternally
imprinted genes are present in dermoid tumors of the ovary, resulting in
development of disorganized fetal tissues without any supporting placenta or
fetal membranes. Conversely, two sets of paternally imprinted genes are present
in hydatidiform moles. In these cases, dysplastic
trophoblast develops, but a fetus does not.



.webp)
_Page_049.jpg)
