Iron Refractory Iron
Deficency Anaemia
(IRIDA)
Rare auosomal recessive cases of
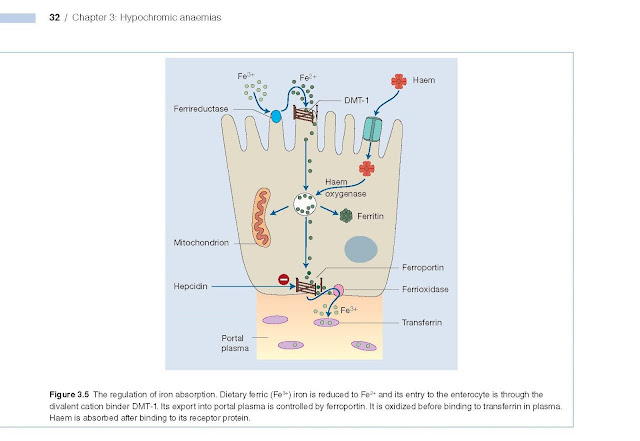
hypochromic microcytic anaemia have been described caused by inherited
mutations of matriptase 2, which allow uninhibited hepcidin secretion, or DMT‐1
genes (Figs 3.4 and 3.5). There may be a haemaological response to
intravenous but usually not to oral iron.



.webp)

