CARDIAC MAGNETIC RESONANCE IMAGING
CMR imaging has continued to advance as a robust cardiac noninvasive imaging technique. Through electromagnetic manipulation of biological hydrogen protons, CMR provides assessment of cardiac structure, function, perfusion, tissue characterization, blood flow velocity, cardiac masses, valvular heart disease, pericardial disease, and vascular disease. Continued improvements in hardware and pulse sequence design have allowed for improved image quality, speed of data acquisition, and reliability, further increasing the usefulness of CMR for clinical applications. CMR is similar to echocardiography in that neither uses ionizing radiation to acquire high-resolution images, which avoids the exposures inherent in invasive coronary angiography and SPECT imaging. CMR offers viewing cardiac motion in any view. In addition, the versatility of CMR permits imaging of a large field of view in nearly any plane, which allows for the assessment of both cardiac and noncardiac pathologies.
Technology of Cardiac Magnetic Resonance
MRI (including CMR) is based upon the electromagnetic manipulation of
biological hydrogen protons. Hydrogen is the most abundant element present
within the human body; it is present in all tissues, whether in water, adipose
tissue, or soft tissue. Each water molecule contains two hydrogen nuclei with a
single proton, and they behave like tiny magnets. Proton spins can be aligned
by application of a powerful magnetic field in the β(0) direction, because of
the appropriate frequency via the Larmor equation (f = γβ, where f is the
precessional frequency; β is the magnet field strength; and γ is the
gyromagnetic ratio). A second radio-frequency electromagnetic field can then be
briefly applied and then rapidly discontinued. As protons return to their
original alignment after the electromagnetic field is turned off
(“relaxation”), they generate a net magnetization that decays to its former
position with energy loss in the form of a radio signal that can be detected
with a radiofrequency antenna and quantified. Image tissue contrast depends on
differences in the decay of net magnetization in the longitudinal plane (T1)
and transverse plane (T2). Through the application of additional
electro magnetic fields (gradient
fields), radio waves coming from the body can be spatially encoded, which
allows localization within an imaging plane.
Data Acquisition Sequences and Techniques
CMR uses two basic imaging sequences: spin echo (“dark blood”) and
gradient echo (“bright blood”). Spin-echo sequences are commonly used for
multislice anatomic imaging, providing clear delineation of the mediastinum,
cardiac chambers, and great vessels. Alternatively, gradient echo sequences are
used more often for physiological assessment of function through cine
acquisitions. Because of higher possible imaging speeds, gradient echo is more
appropriately used for ventricular function and myocardial perfusion
assessment, as well as valvular assessment. Phase contrast imaging (PCI) allows
quantitative flow velocity and volume flow assessment. All cardiac and most
vascular CMR sequences require cardiac ECG gating. Through data acquisition of
segments at different phases of the cardiac cycle, a cine image loop can be
created to track cardiac motion. Perfusion imaging, through the use of
intravenous contrast agents, permits assessment of tissue vascularity. In the
case of vasodilator stress perfusion imaging, assessment of myocardial ischemia
is possible (Fig. 11.5). Inotropic stress imaging, typically with intravenous
dobutamine, allows assessment of new regional wall motion abnormalities.
Gadolinium-based contrast agents, chelated to other nontoxic molecules for
clinical use, are commonly used for imaging the cardiovascular system.
 |
FIG 11.5 Cardiac magnetic resonance stress perfusion imaging,
demonstrating inferolateral ischemia.
Clinical Indications
Ventricular Function
CMR is highly accurate and reproducible, providing clinically useful
measurements of cardiac wall thickness, chamber volumes, and systolic contractile
function (Fig. 11.6). CMR is recognized as the gold standard for assessment of
left and right ventricular function. Left ventricular ejection fraction, left
ventricular end-diastolic volume, left ventricular end-systolic volume, stroke
volume, cardiac output, and left ventricular mass can all be reliably
quantified. Left ventricular diastolic function can also be reliably
interrogated using PCI.
 |
FIG 11.6 MRI can generate images of the heart in multiple user-defined orientation. LVOT, Left ventricular outflow
tract.
Aortic Disease
CMR has rapidly evolved into a clinically reliable, reproducible modality
to evaluate the aorta and its primary branch vessels. Gadolinium-enhanced,
three-dimensional CMR angiography is an extremely rapid technique that can accurately depict aortic pathology. Serial
monitoring of chronic aortopathy can
be monitored safely, without continued radiation exposure, with CMR
angiography.
CMR is the most sensitive cardiac imaging modality for assessment of
myocardial viability and the extent of myocardial infarction. It is the imaging
modality of choice for patients in whom there is a question about whether the
myocardial tissue in the distribution of a planned revascularization is viable
(Fig. 11.7). For this application, compared with nuclear imaging, CMR is much
more sensitive in detecting subendocardial viability (and lack of viability),
and obviously, CMR does not require radiation exposure for patients. Gadolinium
is excluded from intact myocardial cell membranes and thus is useful in
defining areas of infarction. Correlation with anatomic specimens suggests a
sensitivity and specificity of >95%. Delayed hyperenhancement (DHE)
protocols, which most often use phase sensitive inversion recovery imaging, are
based on the high-signal intensity (bright) that results from T1 time
shortening due to gadolinium contrast localization within scar tissue.
Alternatively, first-pass perfusion images that appear hypointense are probably
a combination of ischemic and infarcted tissues. The highest likelihood of
recovery of contractility impairment exists when the transmural infarction extent,
as assessed by DHE, is <50% transmural.
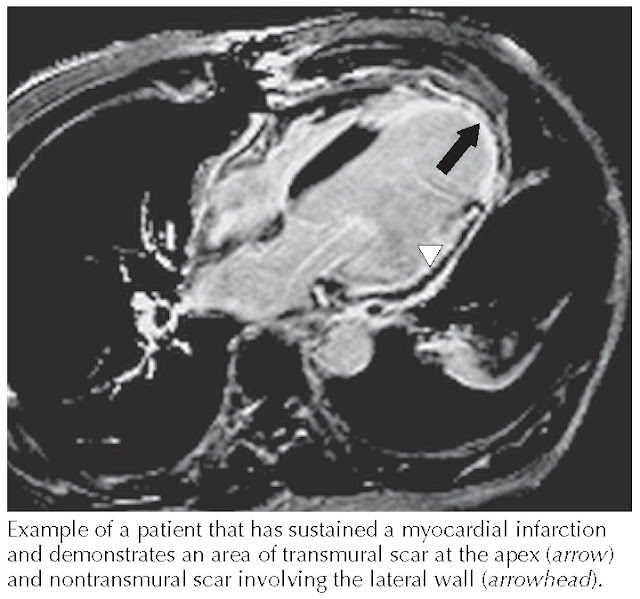 |
| FIG 11.7 Cardiac magnetic resonance imaging: transmural and nontransmural scars. |
CMR is an important tool in the evaluation of dilated cardiomyopathy, hypertrophic cardiomyopathy, and
infiltrative disorders. It provides accurate
assessment of ventricular function in patients with dilated cardiomyopathies. DHE CMR has a niche role
in helping to differentiate heart failure related to dilated cardiomyopathy
from CAD, although the distinction is not perfect. More than 10% of patients
with dilated nonischemic cardiomyopathy have gadolinium enhancement that is
identical in appearance to that seen in patients with CAD.
In hypertrophic cardiomyopathy, CMR can accurately localize hypertrophy, particularly when echocardiographic data are equivocal. Cine images can also demonstrate systolic
anterior motion of the anterior mitral
valve leaflet and dynamic outflow tract obstruction, which are useful measures
in selecting an optimal therapeutic approach in this patient population. More
recent data indicate that increased DHE scar burden in patients with
hypertrophic cardiomyopathy is correlated with increased risk of arrhythmia or
sudden cardiac death. CMR also has a role in the evaluation of patients with
suspected infiltrative cardiomyopathies. Sarcoidosis is an infiltrative
granulomatous disease pathologically known to nonuniformly involve the
myocardium. This patchy distribution tends to result in a moderate to high
number of false-negative cardiac biopsy results. When an initial biopsy result
is negative in patients with suspected cardiac sarcoidosis, the benefits of
repeated biopsy procedures must be considered because of the risks inherent in
this procedure. CMR DHE imaging can depict areas of interstitial changes and
granulomatous disease (Fig. 11.8). In patients with a
high pretest probability for cardiac sarcoid, CMR can potentially serve as a
reliable screening tool, obviating the need for biopsy, particularly if the
diagnosis of sarcoidosis has been confirmed by biopsy of noncardiac tissue.
Amyloid infiltration in the myocardium may
show diffusely increased signal intensity with DHE imaging sequences. In
addition, the combination of ventricular hypertrophy without ECG concordance,
atrial wall thickening, valve thickening, pericardial and pleural effusion, and
restrictive diastolic filling pattern can collectively raise the clinical
suspicion for infiltrative cardiac amyloidosis. CMR is also capable of
confirming the diagnosis of arrhythmogenic right ventricular dysplasia, a
diagnosis that historically is based on meeting several major and minor
criteria. Use of contrast agents and DHE imaging may permit detection of
fibro-fatty right ventricular free wall infiltration, regional right
ventricular wall motion abnormalities, and assessment of indexed right ventricular
volume, which are observations that increase specificity for this otherwise
difficult diagnosis.
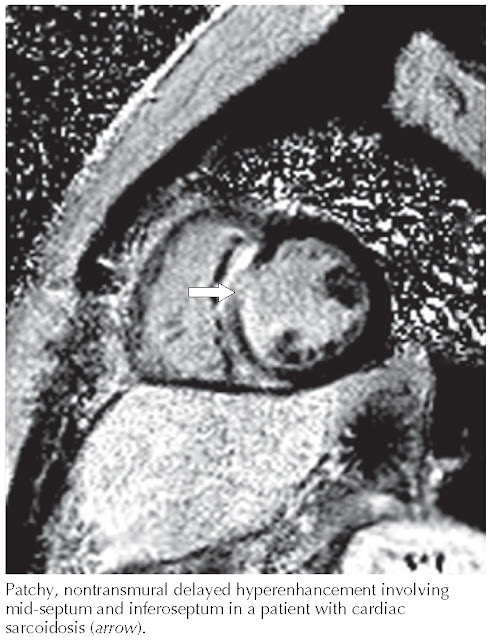 |
FIG 11.8 Sarcoidosis: CMR phase-sensitive inversion recovery.
CMR permits assessment of pericardial effusion, constrictive
pericarditis, pericardial cysts, and
congenital absence of the pericardium. Normal pericardium thickness on CMR is 1 to 4 mm. Functional and
structural abnormalities of the
pericardium can be reliably assessed using CMR imaging. Pericardial DHE imaging
has been demonstrated to correlate with active pericardial inflammation and
neovascularization. In addition, free breathing cine imaging can demonstrate
increased ventricular interdependence suggestive of constrictive pericarditis.
Failure to see slippage between the visceral and parietal pericardial layers
suggests fibrosis, scarring, or connections between these two normally separate
tissue layers. CMR has also proven useful in the evaluation of pericardial
cysts.
Valvular Heart Disease
CMR has become a valuable complementary technique for evaluating the
severity of valvular heart disease. Through a combination of steady-state free
precession and PCI, CMR can provide a comprehensive valvular assessment.
Although echocardiography is capable of superior temporal resolution, is more
accessible, and is less labor-intensive, CMR is capable of imaging flow in
three dimensions (x, y, and z planes), which is more accurate for measuring
absolute flow volumes and feasible in patients whose body habitus precludes
obtaining optimal echocardiographic images. In valvular regurgitant lesions,
PCI can provide exact quantifications of regurgitant volume and fraction. In
patients with aortic stenosis, planimetry of the aortic valve provides accurate
measurements rather than geometric estimations available via echocardiography
and catheterization techniques. In addition, CMR provides accurate measurement
of peak transstenotic jet velocities that are orthogonal to the valve, not merely across it.
CMR is the imaging modality of choice for evaluation of cardiac masses
because of its ability to perform tissue characterization. Spin-echo imaging
provides excellent images for evaluation of the presence, extent, attachment
site, and secondary effects of cardiac mass lesions. CMR has a proven role in
the identification of intracardiac thrombi, primary and secondary cardiac
tumors, and pericardial cysts (Fig. 11.9).
 |
FIG 11.9 Cardiac MRI showing left atrial
myxoma. |
Congenital Heart Disease
CMR is an ideal imaging modality for the assessment of congenital heart disease by providing
superior anatomic imaging coupled with functional interrogation and
reproducibility. In the evaluation of great vessel abnormalities, CMR is the
gold standard, particularly for conditions
such as aortic coarctation. Through velocity mapping of the coarctation jet, a pressure gradient
across the area of narrowing can be determined. Tetralogy of Fallot, including
overriding aorta, membranous ventricular septal defect, right ventricular
hypertrophy, and infundibular or pulmonary stenosis, can be completely
characterized before and after correction. In addition, as is often the case
with patients who required surgical tetralogy repair, CMR is an excellent tool
for monitoring patients for progressive pulmonary valvular regurgitation and
right ventricular dilation. CMR is also capable of reliably depicting anomalous
coronary artery origins and their relation to other cardiac structures and the
great vessels.
Coronary Artery Bypass Graft Imaging
Although coronary angiography remains the gold standard for evaluating
coronary atherosclerotic disease, CMR may be used in the future for noninvasive
assessment of the coronary arteries. The main limitations to CMR coronary
angiography include limited spatial resolution, respiratory motion, rapid
coronary motion (up to 20 cm/s in certain phases), and an inability to easily
assess distal runoff. Quantification (and sometimes even detection) of coronary
luminal stenosis remains challenging. Currently, this is an area of significant
ongoing research. Coronary flow velocities can be estimated by CMR, and some
centers are now using adenosine infusion with CMR to measure coronary flow as a
diagnostic test for functionally important CAD. Anomalous coronary arteries can
be identified using CMR. In particular, CMR is well suited to demonstrate the
relationship of anomalous coronary arteries with other vascular structures (the
aorta and main pulmonary artery), and thus,
to make decisions on the need and timing of surgery.


.webp)

