PERCUTANEOUS NEPHROLITHOTOMY
Percutaneous nephrolithotomy (PCNL) is a minimally invasive procedure for the treatment of kidney stones. In this procedure, a surgical access tract is established between the skin and the renal collecting system. The tract is typically created under fluoroscopic guidance, with needle puncture followed by tract dilation.
Although
more invasive than extracorporeal shock wave lithotripsy (ESWL, see Plate
10-12) and ureteroscopic laser lithotripsy (URSLL, see Plate 10-34), PCNL is
highly effective for large kidney stones and patients with more complex stone
disease. Before the introduction of PCNL, these challenging patients were
managed almost exclusively with open or laparoscopic surgery, which is far more
invasive, requires longer convalescence, and has higher rates of morbidity,
mortality, and stone recurrence. In contemporary practice, however, open
surgery is now performed only in special situations, such as morbid obesity,
numerous stenotic infundibulae, ectopic kidneys without safe percutaneous
access, extremely complex stones that would require numerous access tracts, and
coagulopathies.
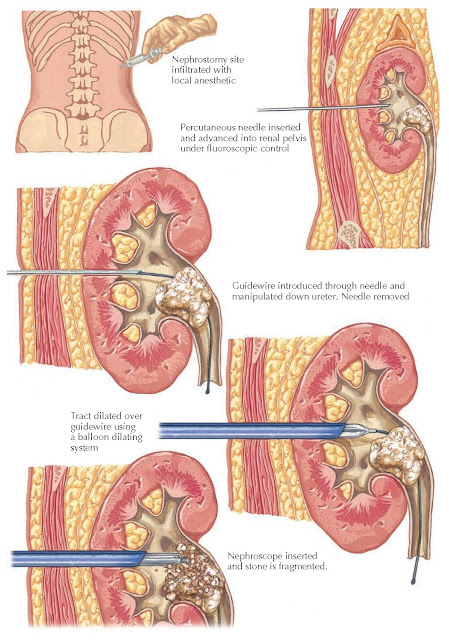
Plate 10-13
PERCUTANEOUS NEPHROLITHOTOMY: CREATION OF ACCESS TRACT
INDICATIONS
The factors
that determine the difficulty, and thus appropriateness, of PCNL include the
degree of hydronephrosis, total stone burden (i.e., surface area), stone
composition, number of calyces involved, presence or absence of infundibular
stenosis, and presence or absence of anatomic abnormalities (e.g., horseshoe
and pelvic kidneys).
The presence
of hydronephrosis makes it easier to maneuver rigid instruments within the
collecting system, and therefore PCNL is preferred over other techniques in
patients with hydronephrotic kidneys.
A large
stone burden is a common indication for PCNL because large fragments can easily
be extracted via single or multiple percutaneous tracts. In contrast, ESWL
requires patients to pass these fragments spontaneously, and URSLL requires
removal of these stones through the ureter.
Stone
composition is an important factor because hard stones, such as those composed
of cystine or calcium oxalate monohydrate, often do not respond to ESWL and are
more difficult to fragment with URSLL. Thus PCNL is usually preferable.
The more
calyces that contain stones, the lower the success rate of ESWL and URSLL,
since it is difficult to treat stones in multiple locations with these
modalities.Thus PCNL
is preferred because of its higher stone-free rate and reduced need for ancillary procedures.
Unusual
collecting system anatomy, in particular infundibular stenosis, often precludes
successful elimination of stone fragments following ESWL and URSLL, again
making PCNL the better choice.
Finally,
congenitally abnormal kidneys, such as horseshoe kidneys and pelvic kidneys,
usually have anteriorly located ureteropelvic junctions, which can make
spontaneous passage of stones after ESWL and ureteral extraction of stones during URSLL very
challenging. The unusual position of the ureteropelvic junction, along with the
collecting system dilation that often accompanies these ectopic kidneys, makes
PCNL the preferred choice.
TECHNIQUE
Patients
scheduled for PCNL must have a documented sterile urine culture because PCNL in
the setting of urinary tract infection
can lead to urosepsis. The procedure is performed under general anesthesia, and
the patient is typically prone. More recently, however, techniques have been
described in which PCNL is performed in the supine and flank positions.
Imaging is
typically performed using fluoroscopy. Before access is attempted, most surgeons
deploy a ureteral catheter into the ipsilateral ureter for retrograde injection
of contrast or irrigant, which facilitates visualization of the renal collecting
system.
The key to
successful PCNL is the site chosen for entry into the collecting system. Lower
pole access is excellent for stones that are in the lower pole or renal pelvis,
but it offers limited access to the other calyces and is thus not well suited
for staghorn stones, complex stones, or stones in multiple calyces. Lower pole
access, however, is associated with a smaller risk of entering the pleural
space. In contrast, upper pole access provides the surgeon the best access to
the entire collecting system, but it is associated with a modest risk of
entering the pleural space.
In most
situations, a posterior calyx should be chosen for access because its
orientation permits entry into the renal pelvis and other calyces from a
typical posterolateral approach. If the patient has a solitary stone, however,
it generally makes most sense to enter the calyx that contains the stone, even
if it is in an anterior position.
Generally,
the posterior upper pole calyx permits maximum stone extraction from a single
access tract.
 |
| Plate 10-14 NEPHROSCOPE AND SONOTRODE |
Once a calyx
has been selected, an important decision must then be made between intercostal
or subcostal placement of the access tract. Subcostal tracts are preferable
because they minimize the chance of pleural injury, which could result in
pneumothorax, hemothorax, or hydrothorax. Such tracts, however, may place a
significant amount of torque on the kidney, potentially causing damage.
In general,
the fear of intercostal tracts is unfounded as long as care is taken to assure
that the sheath remains inside the collecting system. Under these conditions,
most small pleural transgressions will not result in complications. In
contrast, a pleural transgression that is associated with a sheath outside of
the collecting system can result in the passage of irrigant, air, or blood into
the pleural space. A postprocedure chest radiograph or chest fluoroscopy is
recommended to permit early diagnosis of pulmonary complications, so that a
chest tube can be placed if necessary.
Once the
access site has been selected, the procedure is initiated with the percutaneous
deployment of a needle into the kidney. Contrast material is injected through
the needle to confirm appropriate positioning, and a wire can then be placed
into the kidney through the
needle. Using this wire, a tract is developed between the skin and the
collecting system.
In the past,
tract dilation was accomplished using either metallic telescoping dilators (the
Alken system) or Teflon-coated graduated dilators (the Amplatz system). At
present, however, most centers use balloon- dilating systems. These offer
single-step dilation that causes less bleeding than previous systems because of
the application of radial, rather than shear, force. In balloon dilation, a
deflated balloon with a hollow core is passed over the previously deployed wire.
The balloon contains radiopaque markers at its proximal and distal edges to
ensure proper positioning. The balloon is then inflated under pressure. Once the
balloon is turgid, a plastic sheath can be deployed over the balloon. The
balloon is then deflated and removed, while leaving the sheath in position for
continued renal access.
Multiple
access tracts may be required in patients with large or complex stones, such as
staghorn calculi, or with duplicated collecting systems. In some cases, the
need for multiple access tracts can be obviated using the stone push technique,
in which a trocar needle is placed directly onto a stone located in an
inaccessible calyx. The needle is then used to push the stone into the renal
pelvis, from which it can be more easily extracted.
Once access
into the collecting system has been established, the stones are removed. As
most access tracts are 30 Fr in diameter (10 mm), stones up to 10 mm can be
directly grasped and extracted using a flexible nephroscope. Larger stones
require fragmentation (lithotripsy), which can be accomplished using several
different devices. The most common remains percutaneous ultrasonic lithotripsy,
in which a probe with a vibrating tip (known as a sonotrode) produces
ultrasonic energy that fragments the stones. Because the probe is hollow, it
can be attached to suction, such that stone fragments are extracted as they are
created. Pneumatic lithotripsy (hammer-like effect) has also been used for
stone fragmentation alone or in conjunction with ultrasonic stone ablation.
More recently, some authors have described the use of a holmium laser to
fragment stones. Each of these modalities can effectively ablate stones of any
composition; however, ultrasonic lithotripsy remains the gold standard.
After all
stones have been extracted, the kidney is drained to facilitate healing. A
nephrostomy tube can be deployed, but a more recent development is the
“tubeless PCNL,” in which an indwelling ureteral stent is deployed and no
percutaneous tract is left. Although not truly “tubeless,” these stents do not
appear to have higher bleeding rates than conventional nephrostomy drains, even
despite the lack of a large tube to tamponade the access tract.
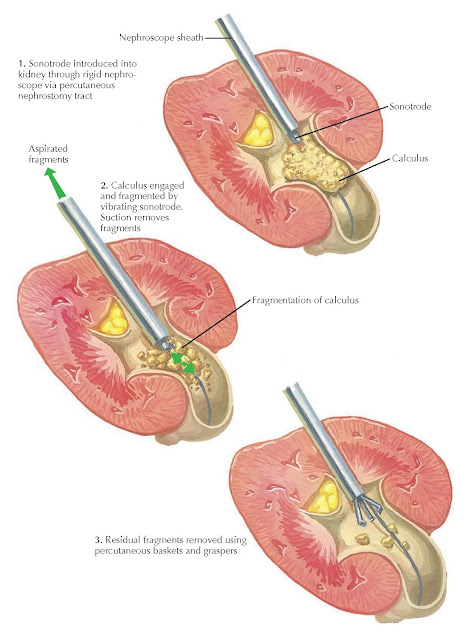
Plate 10-15
ULTRASONIC LITHOTRIPSY OF LARGE STONES
COMPLICATIONS
The major
complications associated with PCNL are hemorrhagic. Occasionally, PCNL procedures need to be aborted before
complete stone ablation if bleeding prohibits adequate stone visualization.
Bleeding is most commonly venous in origin, and it can be controlled by
inflation of the balloon to achieve a tamponade effect. Bright red blood is a
sign of arterial bleeding and should be treated with immediate tamponade
followed by intravascular embolization as needed.
Another
complication of PCNL is perforation of the renal collecting system, which can
result in ascites. In severe
cases, the intraperitoneal fluid can inhibit diaphragmatic contraction and may
necessitate prolonged intubation.
Finally,
residual stones are common after PCNL procedures. Some surgeons perform a
“second look” procedure 1 to 2 days after the primary procedure using the same
access tract. Smaller residual fragments can be treated with active
surveillance, ESWL, or ureteroscopy.




