RENAL REVASCULARIZATION
Renal artery stenosis (RAS) is defined as an anatomic narrowing of the main renal artery or its segmental branches, which can lead to secondary renovascular hypertension (RVH) and renal failure if sufficiently advanced. The pathophysiology and diagnosis of this lesion are demonstrated in Plates 4-36 and 4-37. Briefly, the major causes are atherosclerosis, which accounts for about 90% of cases, and fibromuscular dysplasia (FMD), which accounts for most of the remainder. Atherosclerosis, which tends to occur in older individuals with classic risk factors, involves the intimal layer of the artery and develops circumferentially to occlude a progressive fraction of the vessel lumen. FMD, in contrast, causes collagenous dysplasia of either the intimal or medial arterial layers.
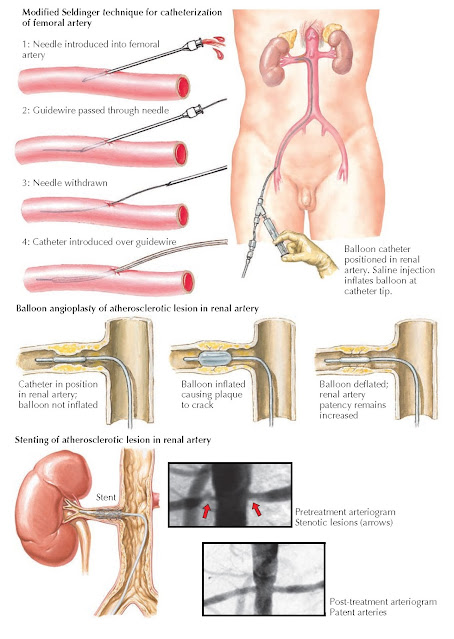
Plate 10-17
ENDOVASCULAR THERAPIES FOR RENAL ARTERY STENOSIS
INDICATIONS
AND EVALUATIONS
The initial
management of RVH secondary to RAS is with antihypertensive medications,
especially ACE inhibitors. The need for surgical intervention depends on the
patient’s response to antihypertensive medications and the degree (if any) of
renal insufficiency. Renal revascularization may be considered if blood pressure
is refractory to treatment with multiple agents, or if patients have bilateral
stenosis or stenosis of a solitary kidney. In the latter group,
revascularization offers the most benefit if there is no intrinsic renal disease
and renal function is either intact or only mildly impaired. The resistive
index, measured on ultrasound, provides some indication of the degree of renal
parenchymal fibrosis, and it has been shown to predict the degree of benefit
following intervention.
When
revascularization is being considered, preoperative assessment depends on the
cause of the vascular lesion. Patients with FMD are generally young and in
otherwise good health. Patients with atherosclerosis, in contrast, are likely
to have disease elsewhere in the vasculature and are thus at increased risk for
postoperative myocardial infarction and/or cerebrovascular accident. Therefore,
such patients should undergo thorough preoperative evaluation, which may
include cardiac stress testing and/or carotid ultrasound.
PROCEDURE
Endovascular
repair has become the preferred method for renal revascularization. Open surgical repair, in contrast, is typically
reserved for patients who have failed endovascular repair, who have
comorbidities such as aortic or renal artery aneurysms, or who have large and
complex lesions.
Endovascular
revascularization consists of percutaneous dilation of the renal artery (often
termed percutaneous transluminal angioplasty, or PTA). The femoral or radial
artery is catheterized using the classic or modified Seldinger technique. Under
fluoroscopic guidance, with
occasional injections of contrast material to opacify the vasculature, a
flexible guidewire is advanced across the stenotic segment of the renal artery.
A balloon catheter is then selected that is approximately equal to the diameter
of the nonstenotic portion of the renal artery. The balloon is placed over the
wire to the level of the lesion and then inflated to a high pressure. In
patients with atherosclerosis, the inflated balloon fractures the plaque,
whereas in patients with FMD, the balloon stretches the vessel wall. In either case, perfusion to the kidney is
markedly improved. A postdilation angiogram is performed to assess the results
and determine the presence of any complications, such as injury to the vessel
wall. An adjunct to PTA is the deployment of an endovascular stent, which is an
expandable, metallic mesh sheath that helps maintain vessel patency. Stents are
especially useful in the treatment of atherosclerotic stenoses, which tend to
be rigid and may recoil after balloon dilation.
Surgical
revascularization consists of bypass of the stenotic lesion or, less commonly,
removal of the obstructing plaque (endarterectomy). Aortorenal bypass is often
performed with an autologous graft, such as the saphenous vein. If an
autologous graft is not available, a synthetic polytetrafluoroethylene (PTFE) or
Dacron graft may be used instead. In patients with severe abdominal aortic
disease, in whom aortorenal bypass would be challenging or even dangerous,
alternatives include splenorenal or hepatorenal bypass. If both the abdominal
aorta and celiac artery have severe stenosis, the lower thoracic aorta may
sometimes be used instead. Simultaneous renal revascularization and replacement
of the abdominal aorta should not be attempted unless there is another
indication for aortic replacement, such as a large aneurysm.
Following
either endovascular or surgical treatment, success is defined as elimination of
the stenotic lesion on postprocedure angiogram or a postoperative blood
pressure of less than 140/90. Many patients show improvements in blood pressure
but do not become completely normotensive. The cure rate is greater among
patients with FMD than among patients with atherosclerosis, in part because the
latter group is more likely to have concomitant essential hypertension.

Plate 10-18
SURGICAL THERAPIES FOR RENAL ARTERY STENOSIS
COMPLICATIONS
After
endovascular repair, patients may experience acute tubular necrosis resulting
from the contrast administered during the procedure. To reduce the probability
of this complication, patients should receive adequate hydration both before
and after the procedure. In addition, all other potentially nephrotoxic
medications should be held. Other complications of endovascular repair include
hematoma formation near the puncture site, thrombosis of the renal artery
secondary to balloon trauma or to inadequate anticoagulation following stent
deployment, and restenosis of the repaired lesion. The most serious complication is perforation of the renal
artery, which is typically noted during the procedure. In this case, the
balloon should be reinflated to tamponade the artery. Emergency open repair may
be necessary if bleeding is persistent.
After
surgical revascularization, complications include persistent stenosis, graft
thrombosis, and restenosis of the repaired lesion. Mortality rates are very low
among patients with FMD, owing to their young age and generally good health,
but range from 2% to 6% among patients with atherosclerosis.
Patients who
have recurrent stenosis after endovascular repair often require surgical
revascularization. The surgical approach may be more challenging because of
perivascular inflammation associated with the initial endovascular procedure;
however, this difference does not appear to lower the probability of a
successful outcome. Patients with recurrent stenosis after an initial surgical
revascularization may undergo another surgical procedure with an alternative
bypass route.


.webp)
_Page_049.jpg)
