MENOPAUSE
Menopause is the loss of normal ovarian steroidogenesis because of age, chemotherapy (alkylating agents), radiation, or surgical therapy. (Menopause may be viewed as an endocrinopathy: the loss of an endocrine function with adverse health consequences.) Menopause naturally occurs at a median age of 51.5, with 95% of women going through this transition between ages 44 and 55. Menopause may occur at a younger age in smokers, those with poor nutrition or chronic illness, or those who have a loss of genetic material from the long arm of the X chromosome.
When ovarian steroidogenesis is lost, menstrual function (if the uterus
is present) ceases. Up to 85% of women will also experience hot flashes, flushes,
and night sweats, with the most severe symptoms associated with the steepest or
most abrupt declines in hormone levels (e.g., surgical menopause). Vaginal
atrophy, vulvodynia, dysuria, urinary urgency, and urgency incontinence,
urinary frequency, nocturia, and an increased incidence of stress urinary
incontinence are also common with the loss of estrogen. For many women, there
is a decrease in libido, independent of that caused by vaginal dryness and the
attendant dyspareunia. Around the time of menopause, there is an
estrogendependent accelerated loss of bone mass. There is a suggestion of an
increased risk of cardiovascular disease associated with natural menopause and
strong evidence of this in premature surgical menopause.
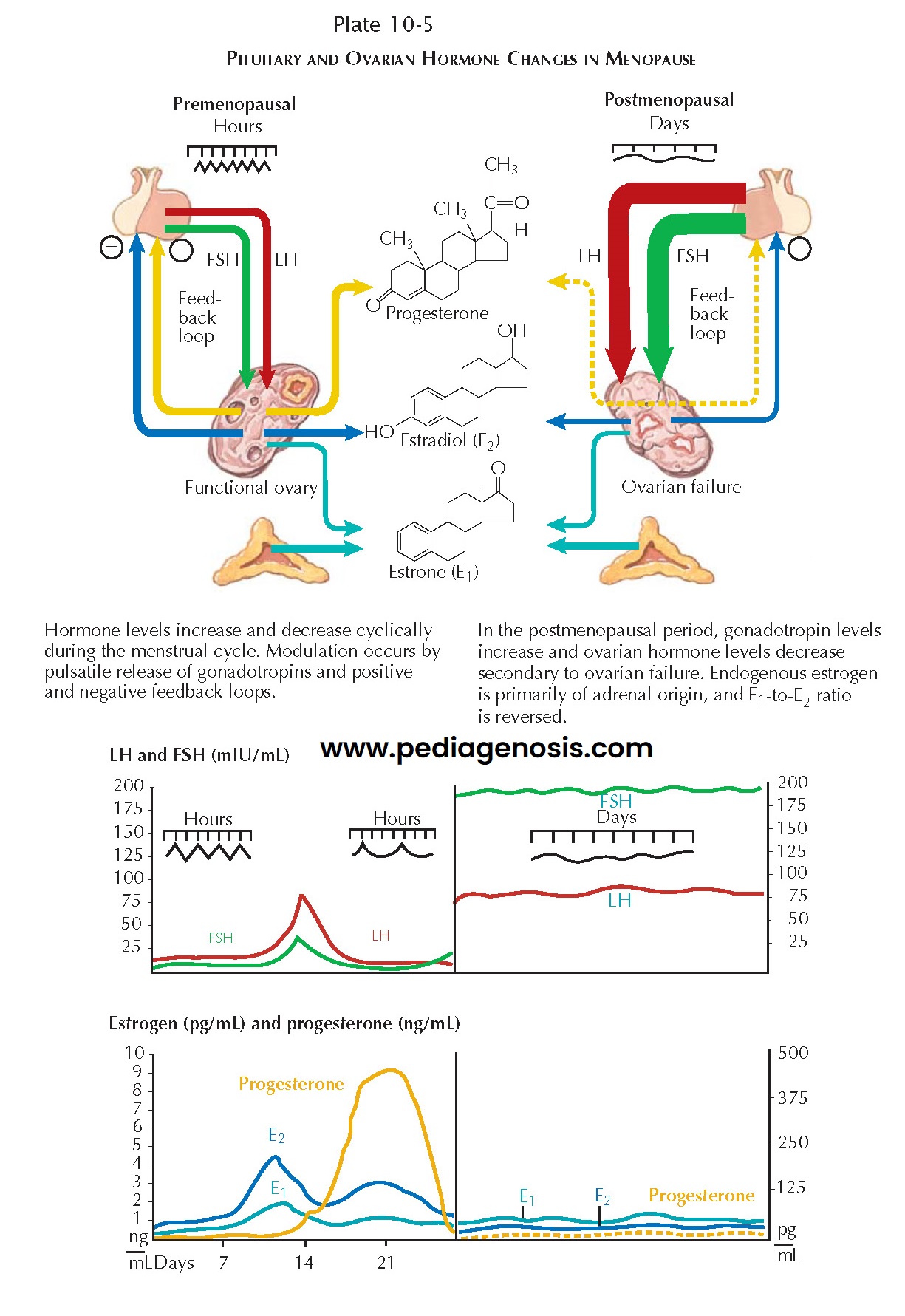
During the natural transition from ovulatory func- tion to postmenopause
ovarian activity (the “climacteric period”), many women will experience
irregular vaginal bleeding and may experience the beginnings of hot flashes or
flushes. Following menopause, the ovary is not truly quiescent: luteinizing
hormone (LH) stimulation of theca cell islands in the ovarian stroma results in
testosterone and androstenedione production. Although these are produced at a
much lower level than before menopause, androgens become the primary hormonal
product of the ovaries.
Although the timing and symptoms of menopause are sufficiently
characteristic to allow a diagnosis to be made by history and physical findings
alone, when the symptoms are atypical or the timing other than expected,
alternative causes for the symptoms such as pregnancy, hypothyroidism,
polycystic ovary syndrome (PCOS), a prolactin-secreting tumor, or hypothalamic
dysfunction should all be considered. When the diagnosis of ovarian failure
must be confirmed, measurement of serum follicle-stimulating hormone (FSH) can
help support the diagnosis. FSH levels higher than 100 mIU/mL are diagnostic.
Although lower levels (40 to 50 mIU/mL) may be sufficient to establish a
diagnosis when symptoms are also present, a single measure of an elevation at
these lower levels is not a reliable indicator of menopause. Serum estradiol
levels may be determined (generally less than 15 pg/mL) but are less reliable
as a marker of ovarian failure. A pregnancy test is always indicated in
sexually active perimenopausal women who are not using contraception. A vaginal
maturation index may be obtained but is generally not required for diagnosis.
Bone densitometry may be indicated for those at special risk for bone loss.
When noncyclic bleeding occurs in these patients, pelvic examination, Pap
smear, and endometrial biopsy should be strongly considered. Women who have
ovarian failure below age 30 should have a karyotype performed.
The management of menopause and its symptoms has become controversial in recent years. Estrogen replacement therapy is still indicated when symptoms such as vasomotor or urogenital symptoms warrant, but most suggest that this therapy should be time-limited. Hormone replacement therapy targeted primarily toward the prevention of bone loss or to reduce the risk of heart disease has generally been replaced by more specific osteoporosis therapies and cardiac risk–reduction strategies. When estrogens are used, progestins are usually added to the regimen if the patient has a uterus present to reduce the risk of endometrial hyperplasia or cancer. (Continuous estrogen exposure without periodic or concomitant progestins increases the risk of endometrial carcinoma six- to eightfold.)