Opioid
Withdrawal
An
opioid withdrawal syndrome (OWS) occurs when an individual, physiologically
dependent on opioids (either due to chronic opioid analgesic treatment or
opioid use disorder), experiences a period of reduced dosage or abstinence from
opioid-taking. OWS is both physically aversive and powerfully anxiogenic; thus
individuals with moderate-to-severe OWS are highly motivated to seek opioid
sources for immediate relief. Pathophysiology. Opioid tolerance
and withdrawal occur as neuroadaptations to chronic opioid exposure. This
neurobiology is complex, involving adaptations at all levels of
opioid-sensitive brain signaling, including (1) mu-opiate receptor desensitization,
(2) opioid-sensitive neuron cellular tolerance due to up-regulation of
adenylyl cyclase activity and changes to cyclic adenosine monophosphate (cAMP)
response element-binding (CREB) signaling, (3) system feedback adaptations
of neuronal and glial networks interacting with opioid-sensitive neurons, (4) opioid-sensitive neural
circuit changes in synaptic plasticity. Clinically, opioid signaling is
inhibitory in function (e.g., suppression of pain, respiratory drive,
arousal, and anxiety); in contrast OWS symptoms are mediated by rebound
hyperactivity due to reduction or removal of chronic opioid agonism.
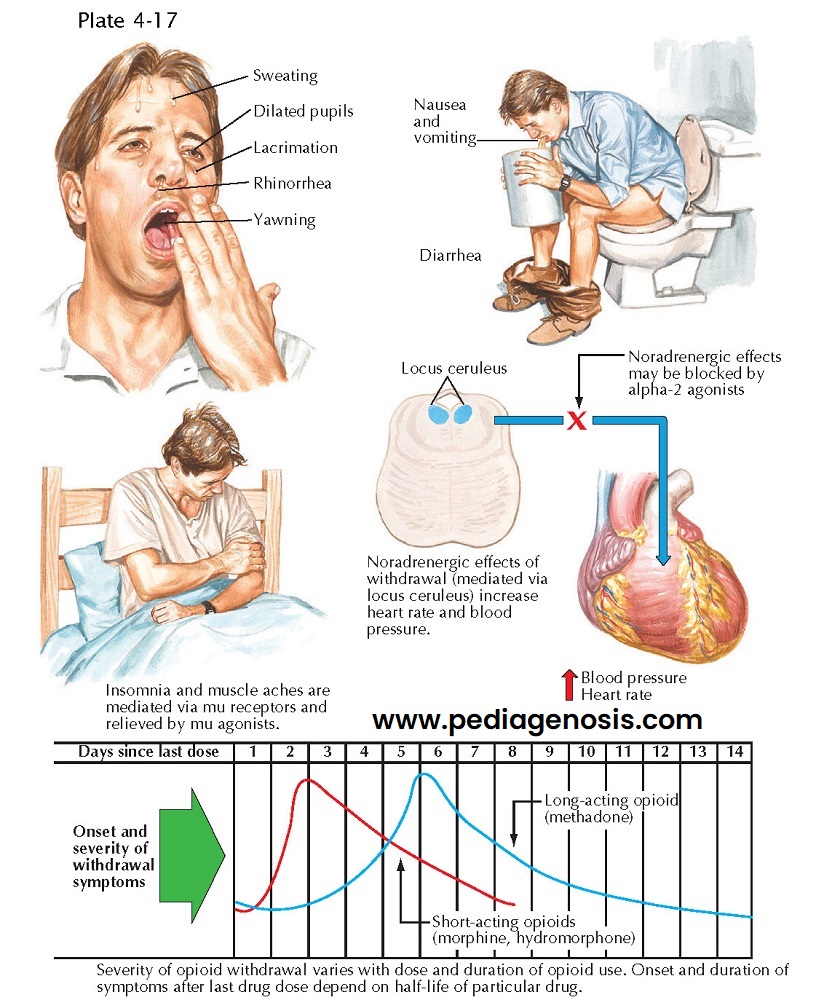
Presentation. OWS onset, duration, and severity vary according to type of opioid exposure (short-acting vs. long-acting, full agonist vs. partial agonist), exposure duration, dosing of exposure, and periodicity of with drawal episodes. OWS is more severe with high-dose, full-agonist opioid exposure; more frequent OWS episodes worsen future withdrawal episodes. Individuals using short-acting opioids (heroin, oxycodone, or hydrocodone) experience mild-to-moderate OWS within 8 to 12 hours of last dosing, whereas persons using long-acting opioids (sustained-release oxycodone or methadone) experience mild-to-moderate OWS within 24 to 36 hours of last dosing. The duration of OWS is briefer (days) with shorter-acting opioids and may persist for weeks with longer-acting opioids. In both instances, protracted withdrawal may persist for weeks to months characterized by residual dysphoria with persistent physical discomfort often interfering with an individual’s motivation and capacity for remaining abstinent from opioids. Craving and mental preoccupation associated with opioid addiction is nearly universal, persisting beyond the acute withdrawal episode, leading frequently to opioid use relapse.
Clinical symptoms of OWS
include anxiety, irritability, dysphoria, nausea, anorexia, chills, muscle
aches and cramps, abdominal cramping, opioid craving, head-ache and fatigue.
OWS patients demonstrate mydriasis, lacrimation, diaphoresis, yawning,
piloerection, rhinorrhea, tachycardia, hypertension, fever, diarrhea, vomiting,
insomnia, and restlessness. Individuals vary in their presentation; some
primarily experience gastrointestinal distress, whereas others demonstrate high
anxiety with cardiovascular hyperexcitability. Of interest, each individual is
consistent in his or her OWS symptom pattern episode to episode. This proves
helpful for discerning OWS during follow-up.
OWS may be measured using
a standardized instrument such as the Clinical Opiate Withdrawal Scale (COWS).
This proves useful for medical documentation and opioid dosing needs
assessment.
Treatment. Typically OWS is not considered a
medical emergency; however, special circumstances may constitute medical
emergencies. These include pregnancy
(deleterious cardiovascular effects on the fetus and third trimester premature
labor risk), cardiovascular disease, disorders involving autonomic instability,
and vulnerability to dehydration. Optimal treatment includes rapid symptom
stabilization to prevent opioid misuse and medical consequences of OWS.
Agonist replacement is the most rapidly effective treatment and is achieved by administering previously used opioids or an agonist substitution therapy, including the full agonist, methadone, or partial agonist, buprenorphine. Full agonists are administered to prevent OWS at any time; however, the partial agonist buprenorphine requires OWS be sufficiently moderate in severity (COWS = 9 or greater) before administration. This avoids inadvertently precipitating severe withdrawal (buprenorphine competes superiorly with full agonists at mu-opiate receptors and thereby has functional antagonist activity in this setting). Typical protocols for first-day dosing of opioid-dependent individuals are 5 to 10 mg oral methadone every 4 hours PRN, not exceeding 40 mg/24 hr, or 2 to 4 mg sublingual buprenorphine every 4 hours PRN, not exceeding 16 mg over 24 hours. Dosing in pregnancy is generally similar, although requirements may be higher during third trimester. Methadone peak and trough monitoring is recommended in pregnant women, concomitant with obstetric consultation. Although detoxification protocols may follow first-day dosing, opioid detoxification generally demonstrates poor out-comes in outpatient settings, with very high rates of recurrent opioid use occurring despite behavioral treatments for opioid dependence. Agonist stabilization or maintenance is frequently preferred to improve long- term stability. Although alpha-2 agonists, including clonidine are widely used to treat OWS, it only ameliorates autonomic symptoms (hypertension and tachycardia) without providing efficacy for other symptoms. Patient comfort and treatment retention with clonidine are poor compared with opioid agonist treatments. During any treatment protocol, other symptom-specific medical adjuncts may be needed, including sedatives for insomnia, antiemetics for nausea, and dicyclomine for abdominal cramping.